Supercapacitors are energy storage devices that have high power density – a thousand times that of electrolytic capacitors typically used in electrical circuits – but low energy density. Energy density is the quantity of energy that can be stored by a device and power density is the rate at which the energy can be released or absorbed by a device.

According to a 2009 Report by market analyst Lux Research, the overall market for supercapacitors is expected to expand from $208m in 2008 to $877m in 2014, a CAGR of 27%. Demand is being fuelled by a growing consumer appetite for increasing functionality in devices such as mobile phones and digital cameras, say the authors of the report, Bridging the gap with supercapacitors: a tale of two markets, with the market split into two segments: consumer electronics and large energy storage applications consisting primarily of transportation and grid power.
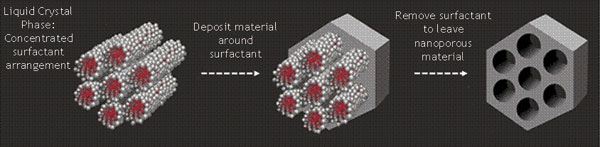

So what does all of this have to do with supercapacitors? Most current supercapacitors feature a symmetrical configuration where both electrodes are made of activated carbon. These devices exhibit good power density characteristics; however, their energy density performance is relatively low, making them unsuitable for many applications. Alternative asymmetrical supercapacitors have a greater energy density, achieved by replacing one of the carbon electrodes with a battery-type electrode that has much greater energy storage capacity. But the downside of this configuration is that the battery electrode responds much more slowly and therefore degrades the power density performance.
Using nanoporous materials to produce the electrodes within supercapacitors means the stored energy is more accessible and can be released faster than in traditional devices. This is because the performance of many electrical storage devices is governed either by the surface area of the electrode or the rate at which charge can penetrate the electrode material. Nanoscale particles have high surface area in a given volume and a small diameter, resulting in easy access to the bulk material. However, the production and handling costs of nanomaterials is typically high. Therefore, the alternative nanoporous approach, which also gives easy access to the bulk material, is a huge advantage.
Nanotecture’s patented asymmetric hybrid supercapacitor comprises a carbon electrode and a nanoporous nickel(II) hydroxide electrode and is capable of discharging up to 75% of its charge in less than 2 seconds. By combining our nanoporous electrode with a carbon counter-electrode we have developed a device that is capable of significantly out-performing standard supercapacitors in terms of both specific energy and specific power. An asymmetric supercapacitor is socalled because there is asymmetry in the capacities of the two electrodes as well as combining two different storage capacities in the same design. The nanoporous positive electrode stores charge like a battery while the carbon negative electrode stores charge like an electrochemical capacitor.
Nanotecture has been able to take advantage of the high energy storage ability of the batteryelectrode while significantly increasing the rate at which the energy can be charged and discharged. In using a high surface area, nanoporous Ni(OH)2 as the positive electrode we have made the solid state diffusion lengths up to 10,000 times shorter than for a standard material. As a result, we have developed a device capable of delivering more energy at higher powers than a standard electrochemical supercapacitor.
Importantly, nickel hydroxide electrochemistry is already well established and is seen as safe and relatively inexpensively to manufacture relative to conventional acid based electrochemistries. The resulting supercapacitor not only has a high energy density, but is able to deliver quick bursts of high power – ideal, for instance, for temporary storage and discharge of electrical energy in hybrid electric vehicles and electric vehicles. It can also be used for energy harvesting, short term power backup for uninterruptable power supply applications and smoothing out short term disruptions in electric power grids among other applications.
The supercapacitor market is driven by growth in automotive and transportation in all forms, renewable energy and industrial applications, such as power management. Conservative estimates suggest that this will be a $1bn market for transportation by 2020. In the automotive sector, we believe that we will see a move away from a single energy source and power delivery system – the fuel tank and internal combustion engine, respectively – towards an integration of several energy sources and power delivery systems that optimise the vehicle power train, for example, batteries, supercapacitors and maybe fuel cells. This approach is already in development and will gain acceptance over the next few years.
Car batteries, for example, have short cycle lives, measured in 1000s to 10,000s cycles, which are often further reduced by high charge/discharge rates. Nanotecture’s supercapacitor can accept high charge/ discharge rates and has a cycle life measured in 100,000s of cycles. By combining these technologies the battery life can be increased and the size reduced. The company is currently exploring a range of manufacturing options for market deployment including pilot line production, sub-contract manufacturing and volume manufacturing. Much will depend on the location and requirements of the end-users. Experience suggests, however, that in the early days of technology roll-out it is best for the company to play the lead role in the manufacturing and distribution effort. Over time we expect to support a range of products each of which is based on the same electrochemistry but ‘sized’ to match the end application.
The improved power and energy density of our material means it can be applied across a wide range of energy storage applications. In many cases the technology can be viewed as complementary to existing battery technology, whereby the battery acts as the main storage device, with the supercapacitor supporting high charge/discharge periods of operation, for example, in cars. Highly efficient regenerative braking and stop/start systems are well suited to the approach.
At least three major markets are immediately available: first, in the transportation sector, particularly those applications associated with energy storage in hybrid vehicles. Applications such as engine stop/ start, regenerative braking and commercial diesel truck ‘cold start’ are placing increasing demands on energy storage devices and are well suited to this technology. Stabilising or load levelling electrical voltages on both cars and commercial vehicles is becoming increasingly important. Having energy stored near onboard end user devices makes it possible to reduce wiring and weight costs.
The second market is in applications related to the electric utility grid. An example application is ‘dynamic sag correction’, which requires that energy be delivered to or extracted from the grid at specific locations to prevent voltage oscillations that may occur when loads are suddenly added or removed. In Uninterruptible Power Supplies (UPS), supercapacitors can be used to solve a reliability problem as they can be sized to deliver the energy required to bridge the time needed to start a back-up diesel generator set. Emerging fast-growth applications will create demand for similar load levelling and storage applications. A third market is energy conservation, such as energy harvesting in elevators or any industrial application that includes repetitive motion and where energy that is currently dissipated as heat can be recovered as electrical energy, similar to regenerative braking in vehicles.
Within the more general energy storage sector, which includes capacitors and batteries, there are a wide range of electrochemistries that could potentially benefit from nanoporous materials. Feasibility studies have shown, for example, that nanoporous material can help to improve the low temperature performance of Li-ion based systems. Yet other potential applications include catalysts, where the high surface area of nanoporous materials could reduce the amount of precious metals required, and sensors, where greater volumes could be absorbed by the capture material.
Manufacturing process
The basic research and technology for nickel hydroxide is complete and a pilot production facility in Southampton Is now being commissioned to produce the nanoporous material in kilogram quantities. This is expected to be producing initial supercapacitor modules in early 2011.
Bill Campbell is ceo of nanotechnology firm Nanotecture, based in Southampton, UK.





