I implore readers to apply for this fellowship or any award through the Society of Chemical Industry (SCI). This award was one of the most impressive that I have come across and SCI’s management team, academic, and professional members are, without doubt, the cream of the crop.
During my time as the SCI Seligman fellow, I was able to attend several meeting and expos to increase my professional foundation and facilitate future collaboration. Firstly, I was able to attend the Foodex show which was held at the NEC in Birmingham where I met the current Chair of SCI’s Food Group, Craig Duckham. Craig and I surveyed the Foodex scene and met with some very interesting manufacturers. It was intriguing from an American food scientist’s perspective to see the similarities and differences in food trends between the two countries.
I was also able to attend the annual meeting for the SCI Food Group and present my findings from the research performed at the University of Birmingham (UoB), alongside several other awardees of SCI grants. The work of the other presenters really impressed me and displayed how powerful these awards from SCI can be.
On a larger platform, I was able to (after the fellowship had ended) present my work at IUFoST 2016. This five-day international congress is renowned for its excellence and provided a perfect platform for disseminating my findings.
During my fellowship time I was able to visit London, Edinburgh, Belfast, Pembroke, and many other places. Without this fellowship I would have never had the time to complete such a fantastic adventure. The friends that I made along the way were ones that I will take with me for the rest of my life. One of the highlights of this adventure was a fishing trip I planned with several of my new and old friends in the Irish Sea. This fellowship was truly one of the best events of my life and I will be forever grateful to SCI for the opportunity. I suggest readers make the effort to apply; you will not regret it.
My research
My research began early in February 2016 when I arrived to begin work under Dr Serafim Bakalis at the in the School of Chemical Engineering at UoB. I spent four months there and returned believing the experience to have been the most influential and positive event in my academic and professional career. Serafim and I have similar minds and it was a great pleasure to work with him and discuss unusual areas of science - doing so on a weekly basis increased my enthusiasm for research.
In my daily professional life as a Staff Research Associate at the Ohio State University, I manage a lab that focuses on industrial cleaning in food processing. Specifically, we focus on optimising cleaning in an effort to minimise water consumption, energy use, and waste from cleaning operations. Dr Bakalis’ lab at UoB focuses on similar concepts but in the personal health care products processing (shampoo, hand soap, mascara etc). That being the case, we naturally leaned towards a research project based around cleaning. The approach became focused in detecting residual material on stainless steel surfaces using instrumentation on the smallest scale because, if we are to reduce water and energy use, we will need better tools for identifying and quantifying the hygiene of a surface.
Title: Identifying and quantifying residual nano-foulant after clean in place (CIP) using atomic force microscopy
In this study, a foulant was developed on a 2cm2 316L stainless steel coupon through a benchtop fouling process. To complete this, a 10% (wt/wt) solution of whey protein concentrate was prepared by blending distilled water and powdered whey protein concentrate. This solution was stirred on a stir plate at room temperature for 1 hour. The stir rate was monitored to minimise foam development. This solution was placed in a standard aluminum cooking pan with a 12cm diameter. Four coupons were placed at each of the four poles of the pan and the solution was heated from 23℃ to 75℃ on a stir plate while being stirred at 300rpm with a 4.5 cm magnetic stir bar. Once the solution reached 75℃ the coupons were either (a) removed immediately or (b) allowed to remain at 75℃ for 1 hour.
To model the removal of foulant material from the surface of the stainless steel coupons, only coupons that were removed immediately after reaching 75℃ in the solution were used. Coupons were exposed to 0.5% (wt/et) NaOH for periods of 1, 15, 30, and 60 seconds, respectively. The surface roughness of the coupons was used as an indicator of residual foulant material.
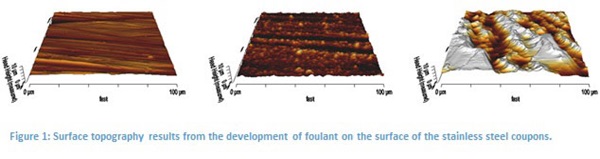
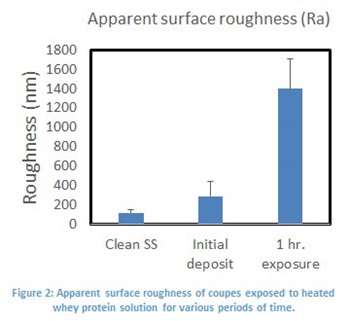
The results for the development of foulant are presented in figure 1. In these images we can clearly see the development of material on the surface. To quantify this effect, surface roughness of the samples was evaluated and plotted against exposure time (Figure 2). These results indicate that surface roughness may be an adequate determinate of residual material on the coupon’s surface.
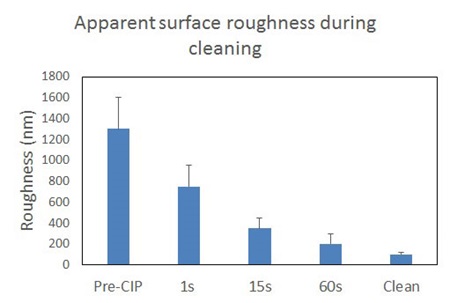
AFM images of the removal of foulant are presented in figure 3. In these images we can see the removal of localized “hills” of what are considered to be residual deposit on the stainless steel surface. To quantify the results, again surface roughness was used to evaluate the removal of these deposits (figure 4)
Conclusion
The research used atomic force microscopy to identify the development and removal of foulant on a surface on the nano-meter scale. Evaluating apparent surface roughness shows promise as an indicator for quantifying residual foulant material. As CIP research progresses, the use of analytical tools to quantify surface hygiene will play a critical role in validating protocols that may be more environmentally friendly.