
Our ability to make particles and coatings with structures the size of biological organelles has opened up new ways to interact with the biological world, resulting in numerous biomedical applications. Think, for example, of magnetic nanoparticles able to bind only to tumour cells in a blood sample and to transport them to a sensor where their concentration can be determined.
Nanocoatings are the key in many of these biomedical applications, be it in vivo, for example in implants, or in vitro, as in devices for blood analysis. Nanocoatings can, for example, make a surface love or hate water, make it repellent for protein binding, transform it into a good environment for living cells, or make it into a binding place for specific molecules.

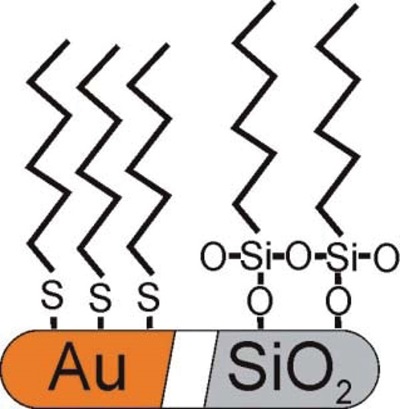
The choice of SAM depends on the nature of the medical device to be coated. In the case of a metal surface, such as gold or silver, alkane thiolbased SAMs are used, whereas for coating oxide surfaces, such as silicon dioxide etc, silane-based SAMs are applied.
Tailored surfaces
Hydrophobic and hydrophilic surfaces: Depending on the medical device application, researchers may choose to make the nanocoating either hydrophobic or hydrophilic. Hydrophobic and hydrophilic coatings are also used in microfluidic circuits in lab-on-a-chip devices.
These coatings can often be produced by linking hydrophilic or hydrophobic functional end groups to the deposited alkane thiol or silane SAM. To make super-hydrophilic or superhydrophobic surfaces, researchers can change their nanostructure. For example, a surface can be made more hydrophobic by increasing the roughness using a method of plasma etching.

Antifouling surfaces: Biofouling is the unwanted adsorption of cells, proteins or DNA onto the surface of a medical implant or biosensor etc. It is a common problem in biosensor applications, where it disturbs the proper functioning of the biosensor and results into noise signal and false positives. One type of antifouling coating applied to biosensors is a protein-resistant coating based on polyethylene oxide (PEO). SAMs with PEO end groups can useful prevent protein adsorption for both metal and oxide surfaces.
Cytophobic and cytophilic surfaces: Yet another area where SAMs may be usefully exploited in biomedicine, meanwhile, is to create surfaces that either attract or repel living cells, termed cytophilic and cytophobic, respectively. For example, bacterial cells grow and even multiply on an agar medium enriched with nutrients, but they will die on a metal surface.
A huge challenge in these neuron-on-chip systems, however, is to make the silicon chip surface cytophilic. Often, poly-L-lysine is used for this purpose. However, Imec has developed a surface-coating technique based on SAMs and a lamininbased protein (PA22-2). The use of SAMs has the advantage that the protein is attached very firmly to the surface, even when a positive or negative voltage is applied to the surface. The PA22-2 peptide binds specifically integrin alpha6beta1, a protein found in the membrane of neurons, which makes the cells adhere very strongly to the chip surface. Also, the PA22-2 peptide promotes the formation from the neuron of outgrowths known as neurites.
Biomimetic surfaces
The foreign body reaction (FBR) is a natural reaction of the body towards foreign objects in the body, characterised by the formation of foreign body giant cells, encapsulation of the foreign object, and chronic inflammation. The functioning of implants can be degraded dramatically by this reaction. For example, electrodes will show an increased impedance and sensors will generate false results due to the fact that the blood around the implant has an abnormal constitution.
Imec’s coatings use soft and sponge-like coatings that mimic human tissue to disguise these inorganic implant surfaces. Hence, the body won’t recognise the implant as being foreign. Biocompatible silicones and polymers can be used. New functionality can also be added to the coatings to further reduce the risk of foreign body reaction (FBR), for example, by adding anti-inflammatory substances.
Low-impedance electrode surfaces: Some in vivo electrodes – for example those used to stimulate or record activity in a specific area of the brain – must be very small. However, this miniaturisation impairs performance by increasing electrical impedance. Electrochemically deposited coatings poly(3,4-ethylenedioxythiophene) (PEDOT) or iridium oxide can help to increase both the conductivity and the effective surface area of the electrode by roughening the surface.
Bio-functionalised surfaces
Nanoparticles are of growing importance in the medical world. They can be used for medical imaging, to transport drugs through the body and even to burn away cancer cells. But the only way they can do this is by bio-functionalising their surface. For example, if you want magnetic nanoparticles to bind only the tumour cells in the bloodstream, you need to coat the nanoparticle surface with a biological receptor that will specifically bind to the tumour cells. These biological receptors can be attached to the nanoparticle surface by SAMs.
Affinity-based biosensors come in many guises, depending on the transducer type: plasmonic biosensors, magnetic biosensors, charge-based and temperature-based biosensors. They all rely on the fact that the target molecule – for example, a marker protein for cancer – must bind specifically to the biosensor surface and that this binding event can be measured, by a change in adsorption peak, temperature etc. In order to achieve binding of the target molecule to the biosensor surface, receptor biomolecules must be immobilised on the biosensor surface. Again, an efficient way to do this is by using SAMs. Imec has developed different SAM-based methods to immobilise antibodies, DNA and other bio-entities on biosensor surfaces, particularly gold and metal oxides.
Mixed alkane thiol SAMs comprise alkane thiols of different length and functionality. Long alkane thiols with functional end groups can be used to covalently bind to the receptor biomolecule, while short alkane thiols can be used to make the SAM layer more stable. Also, the short alkane thiols can be used to prevent the non-specific adsorption of bio-entities other than the target molecules, by using end groups such as PEO. This is important when analysing complex samples, such as blood or urine.
Contrary to mixed SAMs, pre-activated SAMs have the advantage that biomolecules can be coupled to the surface without the need for extra coupling reagents. Imec studies, for example, the use of cyano and N-hydroxysuccinimide groups as an alternative for the commonly used amino, aldehyde and carbonic acid coupling groups.
For example, the antibody for S100betabeta was attached to a cyano-silane SAM. S100betabeta is a marker protein for stroke detection. Another example is the use of a pre-activated N-hydroxysuccinimide PEO-alkane thiol SAM for the detection of the prostate specific antigen (PSA), a marker for prostate cancer.
For most applications, the surface modification consists of two steps: chemical modification of the sensor surface with alkane thiols; and covalent attachment of the receptor biomolecules. For certain applications, however, the direct immobilisation of the receptor biomolecules onto the surface is more appropriate. Moreover, it reduces the number of functionalisation steps. In this direct immobilisation approach, the receptor biomolecules are attached to the alkane thiol during its synthesis, before deposition of the SAM.
Valuable technology
Nanocoatings can be used to influence and even control the interaction between the biological and the non-biological world. Researchers have developed techniques to make surfaces hydrophobic/ hydrophilic, antifouling, cytophobic/cytophilic, resistant to the foreign body reaction, and biofunctionalised. This set of techniques is indispensable for the development of applications such as neurons on chip, biosensors and biomedical implants.
Maaike Op de Beeck is progam manager of the HUMAN++ research program, and Karolien Jans is a project leader involved in interface chemistry for biosensors at imec, based in Leuven, Belgium. imec based in Leuven, Belgium





