Seasonality is a fact-of-life for many businesses, say, ski resorts, amusement parks or ice-cream vans. In a matter of days or even hours, demand can go through the ceiling, or the floor, in response to prevailing weather.
So it is for aircraft de-icing. And last season, 2010-11, went down a chart topper. As Western Europe lay gripped in one of the coldest, snowiest winters in 40 years, consumption of these glycol-based fluids went ballistic. More de-icer was used in the single month of December 2010 than had been delivered over the previous winter’s entire season. Together with transport slowdowns – caused by the same weather – unprecedented demand tested the limits of the supply chain. Although factories, depots and drivers worked around the clock to fill orders, a few airports – such as Berlin and Brussels – reduced operations temporarily, due to a lack of available fluid.
This was not unprecedented. Isolated shortages had been reported in the US in each of the two preceding winters. Still, notes Nigel Westlake, head of aviation sales at Clariant, a leading supplier, ‘Our mission is to keep planes flying, so during the warmer months of 2011, we’ve been double-checking every link of our supply chain to make it extra robust.’ The company has bulked up its transport and storage capacity throughout central Europe, and it has launched a real-time tracking system for customers to monitor their incoming shipments.
No supplier, of course, can completely rule out future shortages. The same phenomenon that creates the business – icy weather – is by definition sometimes unpredictable and always uncontrollable.
Anti-freeze frame
Global air-passenger numbers climb at 3-5% most years, but the deciding figure for de-icer is aircraft movements. Your knees have probably noticed that airlines get better at packing us in their planes, so movements grow more slowly than passenger numbers. According to Airports Council International, these rose about 1%/year globally over the past five years. Long term, say suppliers, aircraft de-icer demand is expected to rise at 2-3%/year.
Precise figures on global volume are not available. Statistics suggest, however, that world consumption of de-icer is somewhere around 180,000t/year in glycol content, which amounts to perhaps 10% of all propylene glycol sales. Major consumers are Europe, North America, Japan and Korea. These countries, which have high flight densities in colder climates, are likely to account for three-quarters of the existing market.
Russia’s intake is not well quantified, but surely sizeable. And, as in so many sectors, China is a wild card. Suppliers clearly have been on their toes to feed growing demand there, but again, precise figures are lacking.
Environmental regulations have long steered de-icer formulations. In the 1980s, when their use truly became obligatory (see Ice-busters – the back story, right), most de-icers were made from ethylene glycol (EG). This was a logical choice, because EG already was a main ingredient of automotive antifreeze, but the workhorse role has since shifted to propylene glycol, due to its lower toxicity. Indeed, propylene glycol sees use in small doses as a moisture absorber in food, cosmetics and medicines.
Depending on whether they are applied as de-icers or anti-icers that prevent ice from returning, fluids vary in their water content and thickness. These are classified as Types I, II, III or IV, with the first and the last seeing the most commercial use.
Additives are dosed to the glycol-water solution in any or all of five types:
- Corrosion and flame inhibitors to protect equipment and to prevent ignition, when in contact with current-carrying metal components;
- pH buffers to boost storage life and heating properties;
- Surfactants to increase wetting;
- Thickeners to raise fluid viscoelasticity; and
- Dye, as an identification aid for users.
Lower carbon
Although additives account for less than 5% of a typical de-icer, they have caused a disproportionate amount of concern among environmental regulators. Particular focus has come on the elimination of tolyltriazoles, which have functioned as corrosion inhibitors and flame retardants.
More recently the spotlight has shone on removing alkylphenolethoxylates as surfactants. Finding the right mix of environmental impact and environmental performance has become a key, if not the key, competitive factor among suppliers. This includes not just elimination of toxicants, but also delivering a lower carbon footprint.
Numerous airports in northwest Europe have begun to take on carbon-reduction goals – either voluntarily or imposed by governments – for their operations, and de-icing often stands out as a target. First, de-icing easily can add thousands of tonnes of carbon dioxide to an airport’s carbon ‘income statement’. Second, removing some of that environmental burden can cost far less than other options. ‘Cutting down on de-icer is one of the more economic ways of lowering an airport’s carbon,’ Andreas Sander, Munich airport’s chief of development and structural design, told the Smart Airports’ seminar in mid-October, 2011. ‘It’s certainly cheaper than another popular option, introduction of electric-powered service vehicles.’
Of course safety cannot be compromised, and room for efficiency hikes is limited, so more than ever, de-icer recycling is mooted as a carbon cutter (See When recycling makes sense, left). The process has been technically and commercially proven at about a dozen airports in the US and about half as many in Europe.
Latest to join the club is Norway’s Oslo Airport, which in autumn 2011 brought onstream a plant to recycle an estimated 700t/year of deicer fluid. The move will spare some 2800t of annual CO2 emissions – mostly indirect. This is roughly equivalent to the emissions of an average new European passenger car being driven for 15m km, or 7400 round-trip flights between London and Oslo.
When recycling makes sense
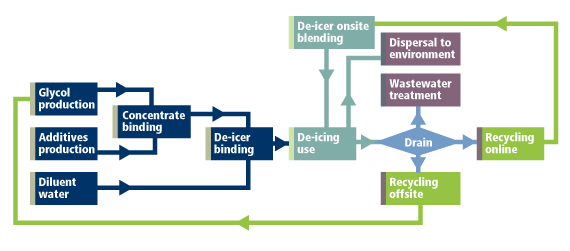
De-icer recycling: Closing the loop cuts carbon emissions
There are plenty of canards about ‘greener’ products. Recycling is not always a good thing, for instance, especially when the recovered material is widely dispersed, either geographically or within other product or waste streams (C&I, 2010, 18, 16).
So, what about aircraft de-icers? When collected by drainage systems adjacent to de-icing pads, de-icers come back in relatively concentrated form. Some 60-80% of the sprayed fluid can be re-captured, most of it at a 10-20% weight concentration amidst snow, ice, rain and water that was sprayed (at point of use, a de-icer typically contains about 40% water). After screening to exclude particles and ion-exchange to remove salts, the spent de-icer can be distilled back to commercial purity.
This final step can take two paths. If done onsite – as at Munich airport – the fluid is purified to 65% glycol and returned to the de-icer supply. If done offsite – as at Oslo and Zurich airports – the fluid is shipped offsite to be purified to 99+% glycol that can see further duty in applications such as heat exchange.
Recycling, rather than discharging to wastewater, cuts the de-icing carbon footprint by 40-50% (Environ. Impact Assess. Rev., 2012, 32, 156). This is mainly because the energy and emissions involved in collection and regeneration are far lower than those created by manufacturing virgin propylene glycol – de-icer’s primary ingredient.
And then there is that other canard: that bio products or processes are always better. Not in this case. Petrochemical-based propylene glycol, used with recycling at the back end, creates a carbon footprint 15-30% lower than that of recently-introduced bio-glycol, made from processing corn.
An extra benefit of recycling has been suggested anecdotally by several airport operators. When decomposing naturally or in wastewater treatment, propylene glycol generates noticeable amounts of methane – a far more potent global warmer than the main breakdown product, carbon dioxide. If collected and distilled for recycling, the spent de-icer has less residence time and availability for methane production.
Ice-busters – the back story
It was one of those public tragedies – like the Piper Alpha explosion, or the King’s Cross underground fire – that triggers changes in policy. In January 1982, an Air Florida flight departed Washington DC’s National airport in the midst of a heavy snowstorm. Ice had accumulated on one of the jet’s external probes, misleading the pilots as to how much power was required for the climb out; extra thrust was needed. More ice and snow on the wings added drag and lowered lift. All this combined to stall the plane, which then crashed into a bridge and the Potomac River, killing most passengers and crew.
The shock was a wake-up call to regulators. Although fluid de-icers had been used since the 1930s and regulations on wing contamination had been established in the 1950s, no widely-followed policy for de-icing existed yet in the US or Europe. Aviation safety experts soon were working on rules: de-icing fluids’ specification, procedures for maintenance, cockpit crews and fluid supply vehicles. De-icing has since become mandatory, and is done according to very detailed, careful guidance.
The use of freeze-point depressant (FPD) fluids such as glycols has remained the most common method for both de-icing and anti-icing. Compared with mechanical and thermal methods, fluid de-icing generally is faster, more flexible and more thorough, bringing maximum safety in minimum time.
Eric Johnson is managing director of Atlantic Consulting, based in Zurich, Switzerland; and Martin Westermaier is technical leader for aviation at Clariant. He is based in Gendorf, southeast Germany.





