Following her insightful SCItalk on Diversity, Equality and Inclusion, Ijeoma Uchegbu, Chair in Pharmaceutical Nanoscience at UCL's School of Pharmacy speaks with Darcy Phillips.
Tell us about your career path to date.
I trained as a pharmacist and then did a PhD at the School of Pharmacy on drug delivery using nanosystems. After about two years of postdoctoral scientist work, I was appointed to a lectureship at the University of Strathclyde in Glasgow. After five and a half years, I was appointed to a professorship in 2002. I then joined the School of Pharmacy as a Professor of Pharmaceutical Nanoscience in 2006 and UCL in 2012.
My work has focused on understanding how drug transport may be controlled in vivo using nanoscience approaches. I co-founded Nanomerics Ltd. with my long-term collaborator – Professor Andreas G. Schätzlein – and next year will see Nanomerics take the technologies developed in academia into clinical testing. This is a huge milestone for our small company and for us personally. I liken this milestone to sending your only child off into the big wide world, and so we are understandably nervous and excited in equal measure!
Science was a refuge for me as I moved countries as a teenager – from London, the city of my birth, to a small town in Nigeria called Owerri. Science subjects were the only subjects that were common on the secondary school curricula of both countries. I really had no other option. I fell in love with science because it was familiar.
Which aspects of your work motivate you the most?
The joy of discovery really gives one a high and this is what I enjoy the most. Validation of one’s discoveries by other members of the scientific community cements the high and when one’s ideas are evidenced first by experimentation and then appreciated by one’s peers, there is no other feeling in the world quite like it.
What has been your proudest achievement?
Getting my professorship so soon after my appointment to a lectureship at the University of Strathclyde is up there with the greatest moments of my career, as is bringing up my daughters at the same time. Oh dear – there are far too many moments to mention, to be honest! Every day I don’t get a rejected paper or grant is really a proud day. Rejections are 90% of a scientist’s life.
You spoke recently at our SCItalk on diversity, equality and inclusion and the importance of data. Could you give us a summary on why this is so important?
To produce good quality science outputs with the maximum impact we need a variety of individuals asking and answering the most profound of research questions. We need more data on diseases and conditions that affect women and more data on the genomics affecting the global southern majority. We need answers to the pressing questions on health outcomes in the poorest in our UK society. Well, you get the picture. We need high quality data on these largely forgotten issues.
What do you think are the next key steps to making STEM more diverse and inclusive?
We first need to recognise that a problem exists. This is the first step. The data on underrepresentation needs to be at the forefront of our thinking when we are making decisions. We need funders to acknowledge the deficit in the current ways of doing things and then commit to act appropriately. The oddest thing about a skewed and unequal system is that we all lose out when there are entrenched inequalities. Even those that think that they are gaining from the current system are not.
We catch up with Zoë Henley, a Medicinal Chemistry Lead at GSK and recent recipient of a Rising Star Award in the category of Science & Engineering
Tell us about your career path to date.
Growing up, I loved science and have early memories of playing with my beloved chemistry set. However, it wasn’t until my A-levels that I knew I wanted to study chemistry at university. I had an inspiring chemistry teacher who supported me to apply, and I ultimately gained a first-class MSci chemistry degree from the University of Bristol.
I joined GSK as an Associate Scientist in 2006 and in my early career I was focused on identifying new molecules to treat respiratory diseases. As a chemist, I use my synthetic and medicinal chemistry skills to identify potential drug candidates, one of which reached Phase 2 clinical trials in patients for the treatment of Chronic Obstructive Pulmonary Disease, which is a life-threatening lung condition with no current cure. I have also led discovery efforts on early-phase research projects to validate the exact role a potential therapeutic target plays in a disease, which is critical for initiating a drug discovery programme.
Through my work, I developed deep technical skills in inhaled drug design and was appointed chair of a technical network for inhaled drug discovery programmes within GSK.
Alongside my work in the laboratory, I also developed my technology skills to become the lead user in Europe for drug compound design and data analysis software.
I was promoted to a Scientific Investigator in 2013 and achieved my PhD in 2014, through a collaborative programme between GSK and the University of Strathclyde. In September 2021, I was promoted to Team Leader.
Over the period I have been a team leader, I have supported 12 scientists and have had the opportunity to mentor graduate chemists and supervise one-year industrial placement students. I am currently a project Medicinal Chemistry Lead and have three direct reports who I support in their professional and scientific development at GSK.

‘I am passionate about science and the job that I do, and am committed to being an advocate for female leaders in chemistry.’ Image: Zoë Henley
Which aspects of your work motivate you the most?
I find the job of a medicinal chemist fascinating and highly rewarding. As a chemist, I have the opportunity to make the molecule that becomes a medicine to help patients, and this is my greatest motivation. Medicinal chemistry is a fast-paced, constantly evolving field that requires diverse skill sets. I find it refreshing to work within a diverse team, in particular working internationally across our global organisation, where I have had experience of working with colleagues across scientific disciplines and from different cultures and backgrounds who bring varied perspectives.
You were recently recognised with a Rising Star Award in the category of Science & Engineering, do you have any advice for anyone starting out their career in this field?
I am passionate about science and the job that I do, and am committed to being an advocate for female leaders in chemistry. For those starting out in this field, I would encourage them to follow their hearts and make well-informed career and personal choices to fulfil their dreams. Whenever I have had decisions to make, I have relied on close friends and mentors for advice, and I would encourage others to identify role models and seek their mentorship. I would also advise pursuing anything you feel passionate about. This might mean, for example, developing a new skill or gaining deeper expertise.
‘GSK as an organisation is highly supportive of flexible working, and within my own department I have continually had support for my professional development, in particular when I returned to work after one year maternity leave.’ Image: Zoë Henley
You’ve been working at GSK for a number of years now – can you tell us a bit more about how they’ve supported you in your career and allowed you to balance this with family life?
Throughout my career at GSK I have had so many opportunities to develop professionally and personally. Alongside continuously developing my technical skills, I have been able to carry out a PhD whilst still a full-time employee of GSK, participated in STEM outreach activities, had supervisory responsibility for both GSK employees and PhD students on collaborative projects, and I have been asked to take leadership roles in many different settings.
In 2019, I became mum to my son Sam, and I have since progressed my career whilst working part-time. I had very few female role models until I came to GSK, where the number of female chemists is high and there were many who had families and successful careers, which gave me confidence that I could have the same.
GSK as an organisation is highly supportive of flexible working, and within my own department I have continually had support for my professional development, in particular when I returned to work after one year maternity leave. My manager was highly supportive of my continued trajectory towards taking a leadership role and supported me in applying for a Deep Dive Career Programme at GSK, which is a competitive programme for future leaders who want to actively shape their career journey.
The programme allowed me to set out a detailed personal development plan and helped to expand my network. The leaders of my department also offered me managerial responsibilities, and this ultimately empowered me to apply for and achieve a Team Leader position.
I have a successful career/family life and aim to give other chemists the confidence that they can achieve the same.
If you'd like to hear more from inspiring female scientists like Zoë take a look at our upcoming SCItalk on Wednesday 27 September: Women in STEM: Better Science and a Better Workplace for Everyone.
CCU International will supply its carbon capture and refinement system to Flue2Chem – a project led by SCI and Unilever which aims to convert industrial waste gases to create more sustainable consumer products. We caught up with CCU International CEO, Beena Sharma, to talk about her career path, motivations and challenges.
Tell us about your career path to date
I joined the Oil & Gas industry after university and began my career as a behavioural safety specialist, specifically for the construction phase of oil and gas projects. Soon after I joined the industry, I was assigned to an LNG plant in Nigeria for training and experience and eventually ended up at a gas plant in Norway before I returned to the UK. With both a psychology and training background I found myself working within a health, safety and environmental remit for various industries including healthcare, construction, manufacturing, and even the tobacco industry.

Beena and colleague at a gas plant in Norway, 2004. Image credit: Beena Sharma
What made you want to work in science and the environmental technology sector in particular?
When I moved to Scotland six years ago it gave me the opportunity to explore the ‘E’ in Health, Safety and Environment further, which was an area that I was always interested in but rarely got the attention it deserved in the industries I worked in. I volunteered on a Scottish climate change project, and this led me to think more deeply about the scientific and technological advances that were needed to achieve net zero by 2045 in Scotland. I knew this was a huge challenge with education, and changes in habit alone could not solve it.
I began to research solutions for hard-to-abate industries and areas that were a challenge to decarbonise, and set up my first business focused on a novel approach to insulating legacy buildings. I then worked on setting up a group of companies that included a solar PV installation company as well as a cleantech business that utilised an electrolysis technology to ozonate tap water for disinfection.
I was invited by my now business partner to help launch a biotechnical business that could create a circular food economy, taking food waste and creating microalgae for use in industries such as cosmetics, pharmaceuticals, and animal feed. This business incorporated 4 technologies, one of which was carbon capture. After some discussion with potential investors, it became clear that there was a huge interest and demand for carbon capture solutions. This led the team to decide to spin out CCU International as a separate entity and speed up the commercialisation of the technology which had been in development at the University of Sheffield under the lead of Peter Styring, Professor of Chemical Engineering and Chemistry.
Which aspects of your work motivate you the most?
The aspects of what I do that motivates me the most is the educational role that I play as the CEO of the business. I am regularly invited to speak on panels, podcasts, webinars and at conferences to share my knowledge with an industry that is transitioning and eager to learn, grow and incorporate new ways of thinking and doing things. It is extremely rewarding to see that people have come away from listening to me with a new perspective and being inspired to go away, take that learning, incorporate it in their ways of working and become innovators themselves.
According to the UN, carbon capture will be a key technology in achieving net zero. It is extremely rewarding to know that the CCU International technology will be a major contributor to this goal and that we can enable decarbonisation with the technology usage across multiple industries, both large and small, which otherwise would not have been possible.
What have been the biggest challenges for you as an entrepreneur?
As an entrepreneur my biggest challenge has been establishing myself in an industry and environment that is not well represented by women, and in particular women of colour. Often, it comes as a surprise to many that I would be heading up such a business and unfortunately many biases still exist within all genders and backgrounds. It makes it that extra bit harder and there can be a requirement to prove oneself as credible through knowledge or capability before the respect is given.

Image credit: Beena Sharma
The other big challenge has been around the education we provide for all our stakeholders. Innovation is not always welcome, especially in an industry or area where it may seem innovation is not needed. As the saying goes, ‘if it’s not broke, don’t fix it’, so stakeholders tend not to realise there is a problem until we educate them on the solution! And not many can accept there may be a better way of doing things than what they themselves have been doing for years!
What would be your top piece of advice for anyone thinking of starting up their own SME?
Starting up in business is a step that many think about doing but very few actually do. Most would be led to believe that you would need to work for months, maybe years on market research, business planning, strategy etc. before starting a business. My one piece of advice would be to start. Most of what you learn will come from doing. It is essential for entrepreneurs to fail, make the mistakes and learn what not to do next time so you have a better chance of success going forward. Many successful businesses emerge from failure.
What is it about the Flue2Chem project that is unique, what made you want to get involved, and what is the potential difference this project could make?
The Flue2Chem project is aimed at converting industrial waste gases into sustainable materials for use in consumer products. What is unique about the Flue2Chem project is that organisations that would normally be competitors have come together to find a solution for a problem that affects us all – as people, as businesses and as a planet. It is rare to see such cross-industry collaboration on this level and this allows both cross-learning and inspires others to come together, collaborate and innovate to solve problems that affect us all, much like the Flue2Chem project. It is a privilege to be part of the project by contributing our technology to the capture component.

CCU International, carbon capture technology. Image credit: Beena Sharma
The project will play a key role in supporting the UK’s 2050 net zero ambitions by providing a more sustainable feedstock for products such as household cleaning materials. The project could demonstrate how the UK could cut 15-20 million tonnes of carbon dioxide emission each year. The UK imports large quantities of carbon containing feedstocks that we use in the consumer goods industry. The project will demonstrate how we can secure an alternative domestic source of carbon for these goods and also demonstrate how industry can contribute towards achieving net zero.
Why do you think collaboration of this scale is so important?
Industry coming together to solve climate change issues is essential if we are ever to achieve net zero. Collaboration of this scale sends a strong message and emphasises that change in approach is needed and that innovation is key. This inspires others to do the same. Solutions are needed now and by bringing expertise and experience together we learn and adapt quicker. Solutions are needed now – not in years to come.
The impact this project will have has the potential to be huge, across multiple industries and certainly with how we look at not only capturing carbon emissions but also what we can do with the captured carbon dioxide, promoting a circular carbon economy where in time we learn to value carbon dioxide in a way that has never been done before.
Certainly, for the carbon capture storage community, this project will show that there is a use for captured carbon dioxide other than treating it as a waste and sequestering in underground oil reservoirs. Utilising captured carbon dioxide can create revenue streams for any business or process that emits carbon dioxide.
The collaboration demonstrates the commitment from industries to support decarbonisation, of those industries that are hard to abate whilst at the same time building a new UK value chain.
Are you interested in pharmaceutical R&D? Which PhD skills are particularly useful in industry? We asked James Douglas, Director of Global High-Throughput Experimentation at AstraZeneca.
Tell us about your career path to date.
I currently have two roles, firstly as Director of Global High-Throughput Experimentation (HTE) within R&D at the pharmaceutical company AstraZeneca. I also work one day a week as a Royal Society Entrepreneur in Residence at the Department of Chemistry in the University of Manchester. Both roles involve developing and applying methods and technology in chemical synthesis to facilitate the drug discovery, development, and manufacturing processes.
My journey to these roles began with a chemistry degree and a passion for running chemistry experiments in the laboratory. At the end of my undergraduate MChem degree at the University of York, I spent an amazing placement year at the pharmaceutical company GlaxoSmithKline, working in drug development. I then went on to do a PhD at the University of St Andrews and postdoctoral research in the USA, both of which focused on developing new methods for synthesis.
My PhD was in collaboration with AstraZeneca and my postdoc was with the pharmaceutical company Eli Lilly, so I knew a lot about medicines R&D and wanted to start a permanent career in that industry.

Pictured above: James Douglas
When did you start working for AstraZeneca?
I started at AstraZeneca in 2015. Initially, I spent most of my time working in the laboratory, supporting drug projects across a range of therapy areas such as oncology, heart disease and respiratory treatment. Since then, I have gradually spent less time in the lab across multiple roles and more time working with – and leading wider teams – with a more company-wide focus.
I have remained closely linked to academic research and universities through collaborative projects. This ultimately led me to the Entrepreneur in Residence role where I am accelerating the translation of chemistry innovation from academia to industry, as well as helping provide students and researchers skills and networks relevant to careers in industry.
What is a typical day like in your job?
I spend about two days a week on site at AstraZeneca in Macclesfield and two working from home. As my main job is office based I can also work from home very easily. It has been this way for me since the start of the pandemic. I missed the general atmosphere of a busy workplace but this period coincided with the birth of my daughter, so I feel lucky to have been able to see a lot more of her growing up than I would have otherwise.
I work with many scientists across the company, not just in Macclesfield, such as in Cambridge (UK), Boston, and Gothenburg, so virtual meetings and calls are a big part of my day. When I’m on site, I prioritise face-to face meetings and discussion with the scientists in the laboratory. Very occasionally I get the chance to run some experiments myself, which I really enjoy.
Since 2022, I have spent Fridays within the Department of Chemistry at the University of Manchester. I talk to academics and PhD students about how their research could be applied in industry, discuss current projects, and think up new ones. I’m also preparing a lecture course and organising careers and networking events to prepare students with skills that are important for careers in industry.
Which aspects of your job do you enjoy the most?
I still get the most excited when faced with the challenge of solving difficult scientific problems. This has changed during my career from working individually in the lab, on relatively clear problems during my PhD, to now being part of much larger teams trying to solve highly complex longer term challenges.
Chemistry is always advancing but so are the standards that we must push towards in drug development – for example, finding ways to shorten the time taken to bring new treatments to patients, while at the same time significantly reducing the environmental impact. That’s a daunting – but exciting – opportunity for synthetic chemists like me.
Most of all, even though the timelines are longer on the projects I work on now, there are moments of short-term success that are exciting. This could be an experimental result from the team that opens up a new possibility, or provides important insight into how best to proceed.
>> Side projects can make large waves. Dr Claire McMullin shares the insights from her journey.
What is the most challenging part of your job?
I miss being able to dedicate my time to experimental work and really understanding a problem in detail. I have spent much of my career investing the large amount of time it takes to understand a problem and think about solutions. Unfortunately, that’s no longer the case and not my main responsibility, but I still find this hard to accept!
I miss the level of detail and discussion I once had and find it a challenge not to spend all my time in the laboratory bothering all the brilliant scientists with questions about what they are doing.
How do you use the skills you obtained during your degree in your job?
Most directly, my degree gave me great general skills in chemistry, ranging from practical experimental techniques to chemical analysis and fundamental principles such as kinetics. These were the basis on which I built more specialised skills in organic synthesis during my PhD and postdoc, all of which are crucial for my career so far.
There are also lots of skills I developed that I didn’t appreciate at the time, such as time management, the ability to think independently, organisation, and teamwork. Like many others, my PhD and postdoc also taught me important lessons about resilience and perseverance.
What advice would you give others interested in pursuing a similar career path?
It’s not advice, but what worked for me was to do what I am passionate about. Don’t worry if it takes a while to work out what that exactly is. I decided to do a chemistry degree mostly because I thought I would enjoy the practical experimental side, which I did and still do. It was only during my final year placement at the pharmaceutical company GSK that I decided to do a PhD so I could learn new areas of chemistry.
Finally, it was only during my postdoc that I decided to try and solve the challenges faced with drug development in industry, rather than the more fundamental undertaken as a research group leader in academia.
I’m still finding out what things interest me and these interests keep changing. That’s the joy of disciplines like chemistry and drug development – there is always so much more to learn and challenges to overcome.
How do you forge a career in process chemistry, and how do you overcome the challenges of studying in your second language? Here’s how Piera Trinchera, Associate Principal Scientist at Pharmaron, found her way.
Tell us about your career path to date.
I am an Associate Principal Scientist in the Process Chemistry department of Pharmaron UK. I am based at the Hoddesdon site in Hertfordshire, where I develop synthetic routes for the manufacture of new drugs for clinical studies.
I’m originally from Italy. I completed my MSci at the University of Salento followed by a PhD in organic chemistry at the University of Bari, focusing on new synthetic methodologies. Despite my complete lack of English at the time, I jumped at the opportunity of a six-month visiting PhD position at the University of Toronto.
This was a challenging experience initially as it was my first time living abroad, but ultimately it was very rewarding. After completing my PhD I returned to the University of Toronto to undertake a postdoctoral position focusing on organoboron chemistry. I followed this with a second postdoc at Queen Mary University of London working on aryne chemistry.
After eight years in academia, I wanted to apply the knowledge I had acquired to solving industrial problems that directly impact people’s lives. For this reason, I joined Pharmaron UK where I have been for the last three years and am currently a project lead and people manager.

What is a typical day like in your job?
I am involved in multiple projects each year and the overall aim is to provide synthetic chemistry solutions for our global clients. Depending on the type of project work, this can include either developing brand new synthetic routes to novel drug candidates or troubleshooting and improving existing chemical processes, making them suitable for large-scale manufacture.
Ultimately, the goal across all projects is the same: to support the production of large quantities of drugs that are needed for clinical studies with a line-of-sight to commercial production.
On a typical working day, I spend the majority of my time in the lab where I conduct my own experiments and lead a team of chemists who work alongside me. I am directly involved in the planning and designing of experiments, execution in the lab, and subsequent manufacture on multi-kg scale in our pilot plant.
Over the course of a project, a large part of the job is communicating to the clients the project strategy, scientific results, and timelines through regular teleconferences, emails, and written reports.
>> Read how side projects made large waves for Dr Claire McMullin
Which aspects of your job do you enjoy the most?
There are many aspects of this job that I enjoy. I have always enjoyed solving new scientific problems, with the thrill of impatiently waiting for the results of an important experiment or the curiosity in trying to understand an unexpected result.
In addition to the science, seeing your day-to-day lab work translated to the production of kg-quantities of new pharmaceutical compounds that might, after clinical studies, further global health is very rewarding.
Projects are completed on much shorter time frames than in academia (three to six months) and there is no time to stagnate as one so often does in a PhD or Postdoc. I enjoy the large breadth in the chemistry and the different challenges that come with each and every project.
Last but not least, it takes many people from different departments (e.g. in analysis, quality assurance, or manufacturing) working closely together to manufacture a drug compound on a kg-scale.
Working so closely with people from different backgrounds has tremendously enriched me during these years in Pharmaron. It has allowed me to acquire new technical knowledge and given me a deeper understanding of not just chemistry but the overall requirements for synthesising pharmaceutical compounds.
What is the most challenging part of your job?
Preparation of a synthetic process for manufacture on a kg-scale involves considerable development in the laboratory to ensure the chemistry translates from small to large scale. Part of this development is to identify potential issues and blindspots of the chemistry and processes and mitigate them by improving the process before implementation on a large scale.
Despite all these efforts, unforeseen complications do occasionally occur on the large scale and finding solutions in real time can be the most challenging aspect of the job. By keeping a clear head, the chemist can leverage both their deep knowledge of the process and the experience of their more senior colleagues to solve these problems.
How do you use the skills you obtained during your PhD and postdocs in your job?
As I’m in a synthetic chemistry job, I have benefitted enormously from the theoretical organic chemistry knowledge and practical laboratory skills that I acquired over the course of my PhD and postdoc years.
Additionally, in academia I became familiar and confident with other skills that I use on a daily basis. These include scientific communication through either written reports or oral presentations, conforming to good laboratory safety practices, and supervising and mentoring other people.
In general, the overall experience of my post-graduate academic education has provided me with the competencies necessary to scientifically manage projects and lead a team in Pharmaron.>> Get involved in the SCI Young Chemists’ Panel.
Which other skills do you need for your work?
Teamwork is a cornerstone of the job and company’s culture. The synthesis of pharmaceutical compounds according to our quality standards would not be possible without the contribution from, and close collaboration among, multiple people across several departments including analytical chemistry, process chemistry, process safety, quality assurance, formulation and manufacturing.
Is there any advice you would give to others interested in pursuing a similar career path?
Don’t be afraid to venture outside of your comfort zone and be open to opportunities, especially those that don’t come along as often. This will help you build your confidence and you will likely find that you can do more than you anticipated. If you are interested in process chemistry, I would recommend looking into internships and/or finding a mentor who can give you an insight into the job.
As with research, perseverance is an important skill you need to master. You will experience failed reactions and difficult purifications at some point in your career as a process chemist. Be open minded, ask questions and don’t be afraid to seek out support from your colleagues.
>> Read how Ofgem’s Dr Chris Unsworth creates an inclusive working environment and transfers his PhD skills.
Do you know how the Academy Awards came to be named the Oscars? What about the story behind the Nobel prize? Behind every award name there is a story, and the Julia Levy Award is no exception.
On the face of it, the Julia Levy Award is about innovation in biomedical applications, but it is the stories of the winners of this SCI Canada award, and Julia Levy herself, that really give it life.
But for a tweak of history, Julia Levy may not have ended up in Canada at all. Born Julia Coppens in Singapore in 1934, she moved to Indonesia in her early childhood. Her father uprooted the family during the Second World War and she left for Vancouver with her mother and sister – her father only joining them after release from a Japanese prisoner-of-war camp.
Julia and her family moved to Vancouver during the Second World War.
After studying bacteriology and immunology at the University of British Columbia (UBC), the young Julia received a PhD in experimental pathology from the University of London. She went on to become a professor at UBC and helped found biopharmaceutical company Quadra Logic Technologies in 1984.
More important than confining her achievements in cold prose, Julia Levy’s work made a profound difference to people’s lives. She developed a groundbreaking photodynamic therapy (PDT) that treated age-related macular degeneration – one of the leading causes of blindness in the elderly. She also created a bladder cancer drug called Photofrin in 1993 and, according to Neil and Susan Bressler, the Visudyne PDT treatment created by Julia and her colleagues was the only proven treatment for certain lesions.
Levy thrived in the business space too, serving as Chief Executive Officer and President of QLT from 1995 to 2001. She has since won a boatload of awards for her achievements, but sometimes the best testimonies come from those who have been inspired by her achievements.
Trailblazing drug discovery systems
For Helen Burt, winner of the 2022 Julia Levy Award and retired Angiotech Professor of Drug Delivery at the University of British Columbia (UBC), Julia has been an inspiration. Here was this UBC professor who jointly founded this big, exciting company – creating medication that improved people’s lives and showing her what was possible.
Helen, an English native, moved to Vancouver in 1976 for her PhD and loved it so much that she stayed. As a professor at UBC, Helen would become a trailblazer in drug delivery systems – a field pioneered earlier by Julia Levy.
‘I was a new assistant professor when she was building Quadra Logic and I would go to talks that she gave,’ Helen said. ‘Essentially, the early technology for QLT was a form of very sophisticated drug delivery [...] It was getting the drug they developed into the eye and irradiating it with light of a specific wavelength.
‘It was very, very targeted. And so, you didn’t get the drug going elsewhere in the body and causing unwanted side effects. So her technology was a form of very advanced drug delivery technology.’

‘For me to win an award that honours Julia Levy and her achievements – I think that's what makes it so special to me.’ – Professor Helen Burt, a former student of Julia Levy, is the Award's most recent recipient.
>> Learn more about SCI Canada.
These talks chimed with the young Helen. If a microbiologist could develop this kind of technology, what was stopping her from developing her own?
She, too, became a pioneer in her field, developing nanoparticle-based drug delivery systems (including those to treat cancer) and a novel drug-eluting coronary stent. According to Professor Laurel Schafer, who put Helen forward for the Julia Levy Award: ‘[Helen] was a trailblazer in new approaches for drug delivery and in research leadership on our campus.’
Importance to Canadian chemistry
Professor Schafer is a hugely accomplished chemist in her own right; and the University of British Columbia chemistry professor’s achievements in catalysis discovery were recognised with the LeSueur Memorial Award at the 2020 Canada Awards.
Julia Levy provided an inspiration to Laurel too, in her case as an exemplar for what Canadian chemists could achieve. ‘The achievements of Julia Levy show that it really can be done right here in Canada, and even right here in British Columbia,’ she said. ‘I grew up in a Canada where I believed that better was elsewhere and our job was to attract better here – a very colonial attitude.

Julia studied at and later became a Professor at the University of British Columbia – the campus is pictured above.
‘I now believe and know that better is right here. Professor Levy’s work showed that world-leading contributions come from UBC and from the laboratories led by women.’
She noted that the Julia Levy Award acknowledges Canadian innovation in health science, whereas Canadian chemistry has historically focused on process chemistry in areas such as mining and petrochemicals.
But Julia Levy’s influence permeates beyond science. ‘Julia is one of those people who has been willing throughout her whole career – even now, well into her eighties – to give back to the community,’ Professor Burt says. ‘She mentors, she coaches, she sits on the boards of startup companies, and she advises.’
‘She’s just got this incredible amount of knowledge… She was the Chief Executive Officer [at QLT], so she learnt all of the aspects: the complex and sophisticated regulations, knowing how to find the right people to conduct clinical trials, and how to do the scale-up. She really is a legend in terms of giving back to the community. And this is not just in British Columbia – it’s Pan-Canadian.’

Pictured above: Julia Levy
For young chemists, the Julia Levy in the Julia Levy Award may just be a name for now, but for those in the Canadian chemical industry and patients all over the world, her influence and her work resonate.
As Professor Helen Burt said: ‘For me to win an award that honours Julia Levy and her achievements – I think that's what makes it so special to me.’
>> For more information on the Canada Awards, go to: https://bit.ly/3VMwNKa
Side projects and small impacts can make large waves. Dr Claire McMullin, Computational Teaching Fellow and Director of Year 1 Studies at the University of Bath, shared insights from her career journey.
What is your job?
I’m a computational chemist, with a focus on inorganic reaction systems and explaining observed experimental trends. I work at the University of Bath, but my job role is a little trickier to answer.
Four days a week, I’m employed as a Teaching Lecturer and the Year 1 Director of Studies. On the fifth day I’m a postdoctoral research assistant (PDRA), overseeing the computational aspects of an Engineering and Physical Sciences Research grant.
Tell us about your career path.
I completed my undergraduate and PhD studies at the University of Bristol – under the supervision of Guy Orpen and Natalie Fey – using crystallography and computational chemistry to investigate organometallic complexes.
I wanted to do a post-doc in the US, so I wrote to a few American computational chemists to see if they had funding or a role available. Luckily one did, and I moved to Denton (University of North Texas) to work with Tom Cundari.
I missed the UK, and so returned a year later to Edinburgh for a three-year post-doc with Stuart Macgregor at Heriot-Watt University in collaboration with Dai Davies at the University of Leicester.
Then I joined Bath, initially as a full-time Teaching Fellow for Computational Chemistry. I was lucky there were computing facilities that had a ‘free queue’ to submit calculations, and I was approached by a new colleague, who asked me if I’d be interested in modelling their reaction systems. I had gained a new side-project and hobby for my evenings.
Eventually, more people asked for me to look at their systems, mostly as the department didn’t have anyone with my specific inorganic and organometallic mechanism skills.
Now, over six years later, I’ve almost finished a three-year grant, published 36 papers, developed connections and external collaborators, and secured more funding to run calculations on our ‘premium’ queue. The only downside is that my research is rarely recognised by the university, as it’s not officially part of the role description of my employment.

Pictured above: Dr Claire McMullin
What is a typical day like in your job?
I tend to get to my office after 8am, and deal with any overnight emails first, before checking our High Throughput Cluster for how my calculations are doing. Teaching begins from 9:15am, and my day tends to be full of meetings (online nowadays), lectures and labs.
Something will always come up that I wasn’t expecting, be it teaching or research related. I always have a page-long to-do list. Normally, I manage to achieve two to three things a day, but almost always end up adding more things to it!
>> Get involved in the SCI Young Chemists’ Panel.
Which aspects of your job do you enjoy most?
I really enjoy the collaborative nature of my work – be it lecturing or teaching a lab to students, seeing a student having that ‘a-ha’ moment, or talking to my colleagues in the department about plans or issues we are trying to resolve.
Similarly, with the research I do, I am often trying to explain someone’s experimental data. I like trying to provide answers or reasons for the chemistry that has occurred. It’s almost like trying to understand a puzzle, and seeing a calculation finished always sparks joy in me!
What is the most challenging part of your job?
The emails, and the tasks and requests they bring, can sometimes derail my entire day (or week).
How do you use the skills you obtained during your degree in your job?
I feel incredibly lucky that, on any given day, I can submit a calculation and use the computational skills I developed during my degree. But I use much more than computational knowledge – doing a degree teaches you to be organised and methodical, as well as how to juggle several tasks at once.
The demonstrations I did as a PhD student are now used daily in labs. The research talks I gave have given me the confidence to stand up in front of a room full of students and lecture them on a range of topics. And the papers and thesis I wrote have given me a keen eye for detail and editing other people’s documents.
>> Read how Ofgem’s Dr Chris Unsworth creates an inclusive working environment and transfers his PhD skills.
Is there any advice you would give to others interested in pursuing a similar career path?
There are so many points where the ‘leaky pipeline’ could have meant I left chemistry and academia. In all honesty, I’m not quite sure how or why I’m still here! [A lot of it is about] luck, being in the right place at the right time, or a job vacancy coming up when you need a new position.
Timing really is key. It’s half-worked out for me. I’m now permanent in my teaching role and still get to run my calculations, which I love; but that often comes at a cost to my own time and is done more as a hobby than something I’m paid to do. It doesn’t work out for everyone, and that is no reflection on their skills or abilities.
I’ve always had back-up plans or ideas if I decided to exit the academic highway. So, if you do want to pursue a career similar to mine, make sure you have something else to fall back on. And just keep working hard, slowly building on the work you want to do. Small impacts can end up making large waves.
In the latest of our Careers for Chemistry Postdocs series, Dr Chris Unsworth, Head of Stakeholder Engagement and Hydrogen at Ofgem, talks about rising to the net zero challenge, creating a productive, inclusive working environment, and transferable PhD skills.
Tell us about your career path to date.
Currently, I’m the Head of Stakeholder Engagement and Hydrogen at Ofgem. Prior to that, I was Private Secretary to the Co-Directors of the Energy Systems Management and Security (ESMS) Directorate at the energy regulator Ofgem. I’ve also worked as Senior Manager in the GB Wholesale Markets team and as a Research & Insight Manager within Ofgem’s Consumer and Behavioural Insights team.

Pictured above: Dr Chris Unsworth
What is a typical day like for you at Ofgem?
I’d say there isn’t a typical day in my job, especially given recent events. Our work needed to shift dramatically to make sure gas and electricity kept flowing at the start of the pandemic and during the sharp increase in wholesale prices for gas.
I wore many hats in my role as Private Secretary. I often acted in a Chief of Staff role for the directorate, getting a sense of the mood within our part of the organisation and advising on how to overcome internal issues as they arise. I also often acted as advisor to the Co-Directors of ESMS as they explored which tools can be used to deliver net zero.
Which aspects of your job do you enjoy the most?
I enjoy being able to work on the net zero challenge in a really meaningful way. I also enjoy being surrounded by colleagues who feel the purpose and weight of responsibility in making progress towards a net zero future. It keeps you accountable, but it’s also really inspiring.
What is the most challenging part of your job?
The reasons I gave above for really enjoying my job can also be described as the most challenging! Delivering a net zero future represents the largest transformation that has ever needed to happen at an industrial level.
Also, because folks are so passionate about their work, it’s really important to make spaces where staff can be transparent and open on their views of the way forward. It’s more important, however, for me to act in a diplomatic manner to make sure we get aligned on a clear and singular route to solving problems.
>> Get involved in the SCI Young Chemists’ Panel.
How do you use the skills you obtained during your degree in your job?
I don’t use the skills I practised in the lab directly in my role. However, there are lots of transferable skills that I picked up from my MChem and PhD in Chemistry. Being able to interrogate evidence and critically assess it is really important in knowing which trends are valid and, therefore, which policy options are the best to investigate further.
Being able to bring data and information from lots of disparate sources and use them to create a clear view of what’s going on is another skill that I practise often. I also do a lot of thinking around systems and flows and the various interactions that go on underneath the surface. Visualising systems and interactions is definitely a helpful skill that I first practised in my degrees.
>> How do you go from a Chemistry degree to a business development specialism? Mark Dodsworth told us his story.
Which other skills are required in the work you do?
My current role is very people oriented and so I need to practise a high level of emotional intelligence. I came out as a gay man while doing my degrees at the University of York and I had specific role models there who helped me explore who I was.
I think my experiences during my degrees really helped grow my capacity for empathy and understanding in others. I’ve been afforded the opportunity to work on a huge number of Diversity & Inclusion initiatives as a result of being open and out at work. I’m also very lucky to work in a space where I feel comfortable to do so.

Pictured above: Dr Chris Unsworth
Is there any advice you would give to others interested in pursuing a similar career path?
If you feel a sense of purpose in something you’re doing, then go in that direction. You will always enjoy your work if you understand why you are doing that work.
This may involve you taking a few left turns as you move between different things, but there’s no need to worry about that so much if there’s a clear and consistent theme and purpose that ties it all together.
How do you go from a Chemistry degree to a business development specialism? We hear Mark Dodsworth’s story.
Tell us about your career path to date.
I graduated from the University of Sheffield with a degree in Chemistry, which included a one-year placement at GSK in Stevenage. Working in heterocyclic chemistry at GSK gave me valuable experience, which ultimately helped me secure my first role in industry.
I joined Vernalis Research in Cambridge as a Synthetic Chemist. After more than five years there, I moved to Manchester to work with the CRUK Drug Discovery team as a Medicinal Chemist.
I am now coming up to three years working for Teledyne ISCO – a US company that specialises in the supply of purification equipment to the scientific community. My job role is Business Development Specialist for the Midlands and Wales.
This job involves focusing on the business growth of Teledyne ISCO products throughout the region with new and existing customers. I also provide ongoing support to our growing customer base, whether that be technical or application related.
What is a typical day like in your job?
Day-to-day, my job role varies significantly, which makes it exciting and dynamic. No day or week is ever the same. It could involve anything from responding to customer enquiries by phone or email, discussions around how our equipment can help with the needs of a group or company, or travelling to a customer to run a demonstration of the equipment.
Installation and training new users is a part of the job that I particularly enjoy. We also do exhibitions, which is a great way to show new customers our equipment, and network with existing customers. Some exhibitions also give us the chance to present to an audience.
Which aspects of your job do you enjoy the most?
A job in business development is so much more than I realised. I’ve always really enjoyed helping people, and this job allows me to do that in so many ways, whether it’s providing equipment that makes the chemist’s life easier and helps them with a problem that they’ve been struggling with, or through application support. I love the networking, getting to know people, and hearing about their work too.
>> How do you forge a career in third-level teaching? See how Dr David Pugh goes about it?

Mark Dodsworth
What is the most challenging part of your job?
Currently the biggest challenge is being at home quite a lot. We can do a lot of support through Zoom, but I’ve missed not seeing our customers and having face-to-face interactions with them.
As part of a sales role, there is a degree of cold-calling required. This is a skill that I didn’t have as a chemist and so I did find it challenging. Ultimately, you are just looking to find those who are interested in your product. A ‘no, thank you’ isn’t anything to be afraid of – you just haven’t found the right customer for you.
How do you use the skills you obtained during your degree in your job?
There are many translational skills that you develop as a chemist and times when these skills come in handy. Presentation skills come in useful when presenting at conferences or to senior management.
Communication skills are important when you are transferring information. Not everyone interprets information the same way, so being clear with the meaning of your words is also important.
Time management and organisation are key to this role too. For example, making customer appointments and allowing time for travel. You also need to make the most of your own time, too, by being organised – for example, seeing multiple customers in one location.
As a result, my calendar is usually planned a month in advance, so organisation skills really help here in the planning of your work.
Is there any advice you would give to others interested in pursuing a similar career path?
This was not a career path I’d ever considered, as I’d always been focused on synthetic chemistry throughout university. The main motivator for me was having the opportunity to work closer with CombiFlash systems, as I’d used these systems throughout my career at GSK, Vernalis and CRUK.
My advice would be to discuss [the roles you are interested in] with as many people currently working in that field as you can. I spent time discussing this kind of role with my friends and networking within the science community before deciding to make the move.
>> Get involved in the SCI Young Chemists’ Panel.
>> Read more about how Rachel Ellis began her career in drug development.
What does an academic’s day look like during term time and in the summer? And how do you get from being a student to teaching at university level? Dr David Pugh, MChem in Chemistry at the University of York, told us about his journey and the skills needed to do his job well.
Dr David Pugh
Tell us about your career path to date.
I look after the delivery of practical chemistry teaching in our undergraduate teaching laboratories in the University of York’s Department of Chemistry. This includes both planning what we are going to teach and teaching students in the lab. I actually came to York for my undergraduate degree and have never left! I completed an MChem degree here, before carrying out a Ph.D here under the supervision of Professor Richard Taylor.
What is a typical day like in your job?
In-term and out-of-term days are like two different jobs. When students are here, the days mostly revolve around delivering teaching in the lab. There are lots of organisational aspects to ensure everything runs smoothly and that everyone (students, demonstrators, technicians etc) knows what’s going on, as well as the teaching.
Out of term time, my job is much more around planning for the future, both the logistics of who’s going to come into the lab when, and the actual teaching content. We’re regularly changing parts of the course, and looking for better approaches with the practical teaching to try to ensure we deliver practicals that are effective in the skills they teach, with the right level of complexity.
>> Interested in a career in chemistry publishing? Then see how Bryden Le Bailly, Senior Editor at Nature, went about it.
So, a day out of term time might see me trying to come up with timetables and planning what goes where, or I might be spending time in the lab trying to develop new practicals or refine existing ones.
Which aspects of your job do you enjoy the most?
Teaching students! This is the most enjoyable part of the job – interacting with the students and seeing them slowly develop their practical abilities. It’s especially nice when you see students you’ve taught from when they arrived at university to studying for a PhD and demonstrating in the labs.
What is the most challenging part of your job?
I find developing new practicals for teaching particularly challenging. When you’re a researcher, the outcome of the practical is the key reason for carrying out the lab work: whether it’s to synthesise a new compound or obtain some data to analyse.
With teaching, it’s different. We’re interested in practical processes and whether they are effective at teaching the students.
Teaching labs have many constraints, and practicals need to be designed to take these into consideration. For example, we think about: reaction times, safety of materials, reaction hazards, new skills introduced, practice at existing skills, costs of materials, equipment availability, how many people could carry out the practical, complexity of any analysis, how the labs relate to theory content, and how long it will take students etc.
Developing new practicals that suit the requirements can be really challenging – and you never know exactly how it will turn out until you run it with students for real.
Dr David Pugh (in the blue coat) with Year 3 students.
How do you use the skills you obtained during your degree in your job?
I think the use of the practical skills I learnt will be self-evident in this job, so I’ll focus on some of the other skills. Communication skills are essential, whether using oral skills to explain subjects to students (individually or in groups), giving presentations (e.g. practical briefings), or using written skills (through the lab scripts).
Troubleshooting instruments is a really valuable skill, as the loss of an instrument could really affect students’ progress on a lab day – so being able to quickly fault find and fix is really useful.
And, of course, the skill of being able to learn something you didn’t know how to do is crucial. Chemistry will keep changing, with new areas coming into existence. For example,. programming and computational chemistry are core components in our undergraduate degree programme now, but I had no previous experience in those areas.
Are there any other skills required in the work you do?
Good IT Skills and administrative skills have proved essential. So much of the successful running of the labs comes down to organisation. Being able to manipulate student lists, experiments, marks, attendance data etc is a crucial part of the role – I’d really struggle without effective database and spreadsheet skills that can quickly and efficiently generate the data I need.
Is there any advice you would give to others pursuing a similar career path?
If you do pursue this career path, make sure you network with others doing the same kind of role. Meeting and discussing teaching approaches with those who can really relate is so useful, and makes you really think about how you design and deliver your teaching.
This became even more useful at the onset of the Covid-19 pandemic, when we met regularly to work together to solve the challenges of practical teaching without labs.
>> Would you like to get involved in the SCI Young Chemists’ Panel? Find out more here.
>> Excited about a career in next generation drug development? Read how Rachel Ellis got involved
Interested in a career in chemistry publishing? Then see how Bryden Le Bailly, Senior Editor at Nature, navigated the path from academia to science communication.
Tell us about your career path to date.
I am a Senior Editor at Nature magazine, overseeing what we publish at the chemistry/biology interface. I completed a MSci in Chemistry at the University of Bristol, followed by a PhD in Organic Chemistry at the University of Manchester in which I looked at signalling with synthetic systems in membranes. I was always interested in education generally, and a great teacher of mine told me Chemistry would have enough to keep me engaged. She wasn’t wrong.
Bryden Le Bailly, Senior Editor at Nature magazine
A short post-doctoral position let me carry on research for a year, but I became more certain that a career in academia wasn’t for me. I enjoyed the idea of research more than its realities, and academia didn’t really work with other life choices I wanted to make. Editorial work suits this balance far better while staying close to the science.
Coupled with my interest in science communication, it looked like a good fit. To read and discuss exciting, cutting-edge research didn’t seem too bad a way to make a living. I looked into editorial jobs and, after discussions with a former editor in the Bristol Chemistry department, I started applying for positions at Nature journals. A locum position at Nature Nanotechnology led to me applying for the permanent position at Nature, where I’ve been for a little over five years.
What is a typical day like in your job?
The core of the job is deciding which submissions to review and publish. So, I read, a lot. The areas I cover comprise how molecules are made and how they can be used to interrogate biology or as therapeutic leads, as well as biochemistry, membrane protein biology, and a few other bits and pieces.
If that sounds like a wide range of topics, it is! It’s the same for all Nature editors. This keeps the job varied and interesting. The rest of the job stems from the papers I handle: overseeing peer review, taking decisions post-review, and what reviewer requests need addressing before we can proceed.
This all involves discussions with my fellow editors. In addition, I speak to Principal Investigators (PIs) and other lab members about work coming out of their labs that might be suitable for Nature.
After we decide we’ll publish something, I look for other ways we can promote the work. I pitch papers we are publishing for associated coverage in News & Views, features, or to go on the magazine cover.
Finally, Nature editors commission reviews and perspectives on topics we think are important and timely, and we discuss with our magazine editors news or topics that we believe should be covered journalistically.
Which aspects of your job do you enjoy the most?
Travelling for the job has to be one of its best perks. I manage to take around five to six trips a year, locally and internationally, to conferences and labs. Discussing brand new science one-on-one with the foremost experts in that field is a massive privilege.
However, I also enjoy supporting early-career researchers to publish in Nature and guiding them through our selection process and expectations. A longer-term way I have looked to support early career researchers (ECRs) is by delivering writing and publishing Masterclasses.
What is the most challenging part of your job?
Saying no to about 90% of what gets sent to my desk at Nature, despite it being (mostly) great science.
>> Excited about a career in next generation drug development? Read how Rachel Ellis became involved in Rachel's Careers for Chemistry blog.
How do you use the skills you obtained during your PhD/Postdoc in your job?
A good knowledge of organic chemistry and chemical biology is very helpful, not only for assessing manuscripts but also to advise on standards for Nature and the rest of the Nature portfolio. I am glad I chose research projects that required me to learn a range of techniques and delve into lots of different areas. Some of the more tangentially related areas to my studies are core responsibilities for me in my job now.
Which other skills are required in the work you do?
An interest in a breadth of science and willingness to learn are key. You will be exposed to areas you had previously never appreciated or knew existed in this job, and it is important to understand every submission from all its angles, and quickly.
This involves effective communication with other editors. Communication and learning skills also come into play when you’re out and about, where you might discuss 15 different subjects over a poster session at the end of a long day, or during a visit to an institute. Finally, editors need a good eye for detail.
Bryden has used his background in organic chemistry to forge a career in publishing.
Is there any advice you would give to others interested in pursuing a similar career path?
Firstly, the pace of the job and its expectations are very different from research. Looking at a manuscript from a scientific and editorial standpoint are two very different things. Consider if you have a critical eye when reviewing papers for a journal or reading the literature.
If you can explain to your colleagues or friends why a piece of research is exciting or ground-breaking, this is a good starting point. However, my principal advice would be to talk to editors.
We go to conferences and are happy to discuss the job in more detail. When I first applied for editorial roles, it was helpful to discuss the position with a former editor. When I didn’t get the jobs I applied for, one of the interviewers called me to explain and encourage me in the right direction. This experience was invaluable in getting me to where I am today.
>> Suze Kundu went from academia to presenting TV shows on the Discovery Channel. Trace her storied career path in Suze's Women in Chem blog.
In the first of our new Careers for Chemistry Postdocs series, Rachel Ellis, Senior Client Proposal Coordinator at drug development company Quotient Sciences, speaks about putting her chemistry skills to the test in a new setting and integrating scientific knowledge with people skills.
Rachel Ellis, Senior Client Proposal Coordinator at Quotient Sciences
Tell us about your career path to date
In my current role as a Senior Client Proposal Coordinator, my primary responsibility is to support the Business Development team by collating technical information from the different business units at Quotient Sciences to prepare proposals that meet the prospective clients’ needs, spanning multiple disciplines of drug development.
I work with subject matter experts in Active Pharmaceutical Ingredient (API) synthesis and scale-up, carbon-14 isotope labelling, formulation development, analytical services and drug product manufacturing to generate complex written proposals for clients looking to accelerate their drug development programmes.
I started my career in chemistry with a Master’s degree from The University of York, which encompassed a year-long industrial placement with a speciality chemicals company in the Netherlands. This was a fantastic opportunity to put my chemistry skills to the test for the first time in an industrial setting and informed my decision to explore a career in chemistry outside of academia.
Following completion of my degree, I started working life as a Research Chemist within a global contract research organisation (CRO). The position was a perfect fit for my interests at the time; it was organic synthesis-focused, within the pharmaceutical sector and involved face-to-face interaction with clients.
After 18 months in the role, I identified my strengths in communication and relationship building so took the decision to pursue a career outside of the laboratory, moving into scientific recruitment where I could apply my scientific knowledge and soft skills in equal measure. I spent four years in scientific recruitment where I developed an array of new skills including networking, negotiating, influencing, account management, people management and performance evaluation.
Following a busy four years, I decided to take some personal time to focus on priorities outside of my career and embarked on a twelve-month career break. This was a fantastic opportunity to reassess my skills, interests and objectives, which ultimately brought me into my current role in proposal development. The position perfectly integrates my scientific knowledge and people skills and offers opportunities for continuous development in a dynamic sector.
What is a typical day like in your job?
A typical day as a Proposal Coordinator involves the evaluation of proposal requests from clients, technical discussions with subject matter experts to define project requirements, the preparation of comprehensive proposals including technical writing, pricing assessments and resource planning and any additional client engagement activities to support the proposal award.
Typically, I would lead the preparation of several proposals at any one given time which may include one or more drug development services.
Rachel Ellis seeks to help deliver life-changing medicines in her current role.
Which aspects of your job do you enjoy the most?
I particularly enjoy engaging with new clients to discuss how we can support them to accelerate the delivery of life-changing medicines to the market with greater speed and efficiency. I also enjoy the diversity of tasks involved in my role (scientific discussions, technical writing, pricing activities and project planning) and the balance between working independently and collaboratively as a team.
What is the most challenging part of your job?
As my role involves supporting multiple proposals at any one given time, time management and prioritisation can be challenging to ensure both internal and external deadlines are met. Organisational skills and open communication are key to ensuring projects are delivered on time and client engagement is maintained.
>> Interested in joining SCI’s Young Chemists’ Panel? Find out more on the Young Chemists Panel's webpage.
How do you use the skills you obtained during your degree in your job?
The breadth of scientific knowledge gained from my degree has provided a robust foundation for my current role and enables my participation in technical discussions across multiple scientific disciplines. Report writing, time management and attention to detail are also key skills that I now apply on a day-to-day basis.
Which other skills are required in the work you do?
My current role requires collaboration between many individuals (both internally and externally) across a multitude of disciplines, including technical experts, project managers, business development teams and financial teams.
Strong interpersonal skills are key to ensuring all parties are engaged and aligned in decision making processes. Effective communication skills are also the foundation for a career within any client-facing environment.
Is there any advice you would give to others interested in pursuing a similar career path?
In general, I would strongly advise investing time to evaluate the variety of roles available within the science sector. Don’t be afraid to explore opportunities outside of the norm. Over the course of my career to date, my eyes have been opened to the breadth of roles available within science that are not necessarily laboratory-based, such as regulatory affairs, quality assurance, medical communications and commercial positions.
I would also advise regular self-evaluation to assess your strengths and areas of interest at any given time to assist in the building of a personalised career development plan. This will help to focus your attention on opportunities to develop the skills you need and seek out exposure to relevant activities either within your current organisation (i.e. attending client calls/visits or developing interpersonal skills through participation in cross-departmental activities) or through voluntary work and networking.
>> Interested in a career in science communication? Read Suze Kundu’s inspiring story.

Sharon Todd, SCI CEO
It’s always great to meet new – and old – contacts at events. For so many months, crossing borders wasn’t possible, physically at least, due to the Covid-19 pandemic. Thankfully, the Chemical Innovation Conference (CIEX) provided a welcome change.
On 6 and 7 October, we came together in Frankfurt to discuss the challenges and opportunities in our sector. It was an honour for me to give the opening address – in the same year as SCI’s 140th anniversary.
Indeed, this year’s event included a well-paced mixture of talks and panel events that addressed post-pandemic difficulties, the challenges of climate change, the need to innovate and much more.
The chemical using industries face an array of challenges besides the practical fallout from the Covid-19 pandemic. Brexit, new regulations, supply chain issues, climate change, sustainability, and geo-political unrest pose significant problems
As an innovation hub, SCI connects industry, academia, patent lawyers, consultants, entrepreneurs and government, and other organisations. And I like to think of CIEX as an innovation hub too.
We have no choice but to innovate, but we must do so in a collaborative, sustainable way. The climate change emergency, for example, means society is looking to chemistry to help find long-term innovative solutions. That’s what made CIEX such an apt time for those in the industry to come together and navigate these challenges.
Innovating beyond barriers
The theme of this year’s event was ‘Game-Changing through Collaboration’. But I also thought of it as Crossing Borders – not just physical borders, but getting through the barriers that block innovation. These barriers hold back the translation of scientific solutions from the laboratory into business and, ultimately, into society.
Our sector is in the spotlight as never before and we can shape a better future. The debates at this year’s CIEX, and the exchange of ideas that took place, will help move us all forward. And what an exchange of ideas it proved to be.
We heard from an amazing line-up of speakers, addressing some of the industry’s most salient issues. BASF’s Christian Beil spoke about how best to leverage lean experimentation and rapid prototyping to improve customer centricity in product design, while Iris AI’s Anita Schjoll Brede described how we can reimagine the R&D work environment.
Ineos Styrolution plans to recycle polystyrene using thermal decomposition or by washing and remelting waste.
Furthermore, Johnson & Johnson’s Luis Allo spoke about the rise of consumer awareness as a driver for innovation. He provided interesting insights on accessing information on real customer trends and needs. Dupont’s Fred Godbille also described several tried and tested methods to assess the voice of the customer.
Elsewhere, Croda’s Nick Challoner assessed how we can unlock innovation through collaboration and partnerships. He also provided an overview of how Croda interacts with universities. On a more technical note, Roman Honeker of Ineos Styrolution outlined the company’s plans to recycle polystyrene using thermal decomposition or by washing and remelting waste.
The discussion on ‘how SMEs interact with corporates’ provided another of the event highlights, with contributions from Clariant, BASF, Chemstars, and SCI’s David Bott. Delegates discussed how SMEs sometimes oversell the potential of their products (without necessarily having much real-world experience) and the allegedly slow-moving, risk-averse nature of some corporates.
Cross-border innovation
Throughout the event, attendees examined what we can do better, how this can be achieved, and the resources needed to make this happen. After all, we must be nimble and flexible in these times of political and social uncertainty.
We can cross borders together – physically and virtually – via close collaboration. And we can cross the borders of what’s possible innovation-wise, removing barriers and journeying into new territory for us all.
To celebrate Black History Month, we take a look back at some of the great Black scientists and innovators. From laser eye surgery to the gas mask, here are some of the seminal contributions made by these ingenious inventors.
[1] Lewis Howard Latimer – Image credit: Unknown author Unknown author, Public domain, via Wikimedia Commons
[2] Leonidas Berry - Image credit: Adundi, CC BY-SA 4.0, https://creativecommons.org/licenses/by-sa/4.0, via Wikimedia Commons
[3] Betty Harris – Image credit: https://www.blackpast.org/african-american-history/harris-betty-wright-1940/ - Fair use image
[4] Patricia Bath - Image credit: National Library of Medicine, Public domain, via Wikimedia Commons
[5] Philip Emeagwali - Image credit: SakaMese, CC BY-SA 4.0, https://creativecommons.org/licenses/by-sa/4.0, via Wikimedia Commons
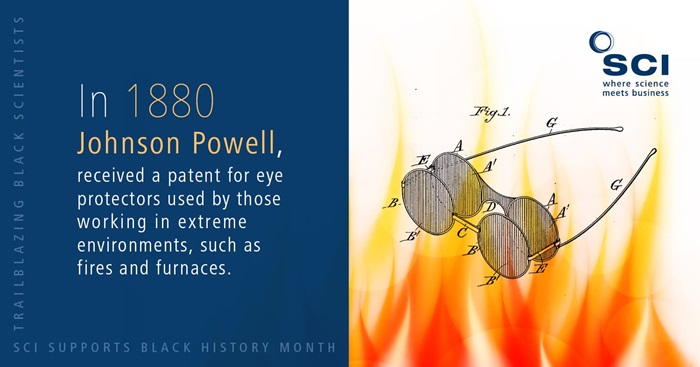
1880 – Johnson Powell
Have you ever used eye protectors to protect yourself against the glare of intense light? For those working in extreme environments such as fires and furnaces, Johnson Powell’s eye protectors will have been a sight for sore eyes.

1881 – James Wormley
James Wormley invented a life-saving apparatus for boats. His contraption included a string of floats that extended from a ship’s side via a sliding rod with projecting arms. The famous hotelier was also said to be at President Abraham Lincoln’s bedside when he died.

Image Credit: Unknown author, Public domain, via Wikimedia Commons
1882 – Lewis Howard Latimer
Lewis Howard Latimer is probably best known for inventing a durable carbon filament that was key to the success of the electric light bulb. Latimer also invented an evaporative air conditioner and even drafted the drawings to secure the patent for Alexander Graham Bell’s little known invention… the telephone.
>> Click here for more on Lewis Howard Latimer’s extraordinary contribution to science.

1912 – Garrett Morgan
Imagine using your own invention to save people’s lives? That’s exactly what Garrett Morgan did when he donned his patented smoke hood to rescue trapped men from a smoke-filled tunnel beneath Lake Erie. Morgan’s device later evolved into a gas mask, and he also invented a three-position traffic signal, hair straightening cream, and a self-extinguishing cigarette for good measure.

1916 – Madeline M. Turner
Madeline M. Turner’s ingenious invention was the fruit of her own frustration. Turner grew tired of squeezing oranges for her glass of juice, so she created the fruit press machine to solve the problem.

1932 – Richard Spikes
It’s safe to say Richard Spikes was a polymath. The American inventor created an automatic gear shift device for cars, a pressurised beer tap, and a horizontally swinging barber’s chair – all while working as a teacher and barber and being a capable pianist and violinist.

Image Credit: Adundi, CC BY-SA 4.0, via Wikimedia Commons
1966 – Leonidas Berry
This doctor and civil rights advocate invented the Eder-Berry gastroscopy endoscope in 1955, which helped doctors to biopsy the inside of the stomach without surgery. According to the US National Library of Medicine, ‘the Eder-Berry biopsy attachment made the gastroscope the first direct-vision suction instrument used for taking tissue samples during gastroscopic examination’.

Image Credit: https://www.blackpast.org/african-american-history/harris-betty-wright-1940/ - fair use image
1984 – Betty Harris
Perhaps the most explosive discovery of all belongs to Betty Harris. Harris’ spot test for detecting 1,3,5-triamino-2,4,6-trinitrobenzene in the field is used by US Homeland Security today to check for nitroaromatic explosives. In her spare time, Harris has even found the time to work with the Girl Scouts to develop a badge based on Chemistry.
>> SCI is proud to support #BlackinChem. Take a look at some of our recent work.

Image Credit: National Library of Medicine, Public domain, via Wikimedia Commons
1988 – Patricia Bath
Patricia Bath has helped return the gift of sight to thousands of people. The US ophthalmologist invented a quick and painless device that dissolves cataracts with a laser and cleans the eye, enabling the simple insertion of a new lens. Her laserphaco probe is still in use today.

Image Credit: Philip Emeagwali - SakaMese, CC BY-SA 4.0, via Wikimedia Commons
1989 – Philip Emeagwali
Nigerian computer scientist Philip Emeagwali won the prestigious1989 Gordon Bell Prize in Price Performance for a high-performance computer application that used computational fluid dynamics in oil-reservoir modelling. In the same year, Emeagwali also claimed to perform the world’s fastest computation – 3.1 billion calculations per second – using just the power of the internet.

2002 – Donald K. Jones
Donald K Jones made a notable contribution to medicine with his invention of a detachable balloon embolisation device that reduces the size of aneurysms (bulges in blood vessels). The endovascular occlusion device is implanted into the body, whereupon its clever balloon system and adhesive materials reduce the size of aneurysms.
>> Which barriers still block the way for Black chemists? Read Claudio Lourenco’s story.
SCI’s America International group has awarded the 2021 Perkin Medal to Dr Jane Frommer. The 114th Perkin Medal was presented to Jane at the Bellevue Hotel in Philadelphia, Pennsylvania, in recognition of her outstanding contribution to chemistry.
Dr Jane Frommer
Dr Frommer is renowned for her key contributions in electronically conducting polymers and scanning probe instrumentation. Her pioneering work with scanning probes paved the way for their use in chemistry, materials science and, eventually, in nanotechnology. According to SCI America, her nanoscopic analytic methods are vital to nanostructural research and are used across many industries.
Dr Frommer began her career in 1980 at Allied Corporate Laboratories (now Honeywell), where she created the solution state of electronically conducting organic polymers. In 1986, she joined IBM where, along with other instrumentalists, she demonstrated the ability to image and manipulate single molecules using scanning tunnelling microscopy. During her multi-year assignment at the University of Basel Physics Institute in the early 1990s, Dr Frommer’s team expanded the capability of scanning probes in measuring the functional properties of organic thin films with atomic force microscopy.
Since 2018, she has worked as a science advisor for Google. In this capacity, she has sought to increase the amount of open source data available in the physical and life sciences. She also helps Silicon Valley start-ups navigate the chemical and material challenges of nanotechnology and has mentored countless students and young scientists in high school, college, and in her laboratory in recent decades.
Previous recipients of the Perkin medal include Barbara Haviland Minor, of the Chemours Company, and Ann E Weber, of Kallyope Inc.
Dr Frommer has written more than 100 referred publications and is the co-inventor of more than 50 issued patents. With her extraordinary body of work spanning more than 40 years, she is a worthy recipient of the prestigious Perkin Medal.
The Perkin Medal is widely acknowledged as the highest honour in American industrial chemistry. It was established to commemorate the 50th anniversary of William Henry Perkin’s discovery of mauveine at the age of just 18. Perkin’s creation of mauveine, the world’s first synthetic aniline dye, revolutionised chemistry and opened up new frontiers in textiles, clothing, and other industries. Perkin was a founding member of SCI and this Medal was first presented to him in New York in 1906.
For more information on the Perkin Medal and the nomination process, visit: soci.org/awards/medals/perkin-medal
SCI was pleased to support #BlackInChem, working alongside our Corporate Partners and members to amplify the voices of our Black chemists.
We have heard stories from several Black chemists who highlighted the steps being taken by many companies to increase diversity. But we can also see that there are many more steps that can be taken to encourage the next generation of budding Black chemists and scientists.
#BlackInChem has had support from Scott Bader, an SCI Corporate Partner, with both Damilola Adebayo and Luyanda Mbongwa sharing their perspectives as employees of Scott Bader. Elsewhere, Cláudio Laurenço gave a compelling account of his journey to become a post-doctoral research associate at a leading consumer goods company.
Cláudio Laurenço worked for free and was overlooked before eventually securing his PhD and starting his career in chemistry.
These chemists are following in the footsteps of some pioneering Black scientists such as Percy Lavone Julian, who has been profiled on the SCI Blog.
Many organisations have expressed their support and shared thoughts on what steps they are taking to encourage and ensure diversity. Indeed, #BlackInChem is a global effort and companies such as GSK have shown their support as well as numerous Black chemists talking about their experiences and achievements over the last week.
Percy Lavon Julian’s pioneering work enabled a step-change in the treatment of glaucoma | Editorial credit: spatuletail / Shutterstock.com
Over the coming months, we will be profiling other Black chemists, past and present, and continuing the dialogue around diversity.
>> We’re always keen to hear about diverse perspectives within chemistry. If you’d like to share your story, please contact: muriel.cozier@soci.org or eoin.redahan@soci.org.
For Cláudio Lourenço, the path from student to multidisciplinary scientist has been far from smooth. The Postdoctoral Research Associate reflects on the institutional challenges that almost made him give up, the mentor whose support was so important, and the barriers that block the way for young Black chemists.
Please give a brief outline of your role.
I work for a leading consumer goods company. I am a multi-disciplinary scientist contributing to the development of novel formulations for household products.
Why are you supporting #BlackInChem?
I’m supporting #BlackInChem because I am a champion for diversity. I believe that what we see from our windows in the street is what we must have inside our workplaces. In an ideal world we should all have the same opportunities, but unfortunately this is somehow far from the truth. We need to motivate our young Black chemists to aim for a career in science by providing welcoming environments and real opportunities instead of just ticking boxes. We need to showcase our Black chemists to show to the younger generation that they can also be one of us.
What was it that led you to study chemistry and ultimately develop a career in this field? Was this your first choice?
I have always been passionate about research and science. My father had a pharmacy, so I was always close to chemistry and was a very curious child. Yes, it was my first choice but the lack of opportunities and trust from universities and scholarship providers made it a long run. My motivation faded and I nearly gave up.
Was there any one person or group of people who had a specific impact on your decision to pursue your career path?
Yes, but after my degree I nearly gave up. It took me nearly two years and changing cities to find something (a voluntary position). I was always keen on taking up mentors to show me how to progress in my career. There were a few people who helped me by training me and teaching me how to navigate the scientific world and pursue a career in science.
I only got my first job (which I worked for free) because of Peter Stambrook, an American scholar from the University of Cincinnati, who I met through a friend while polishing glasses in a restaurant. This man was open and keen to put a word in for me at a leading university in the UK. He taught me so much on how to be a scientist and humbly grow up and make a career in science. Eventually, all his advice kept me on the right path.
What impact would you like to see #BlackInChem have over the coming year?
More Black students in postgraduate courses and an increase in role models to motivate the younger generations to pursue careers in chemistry.
Could you outline the route that you took to get to where you are now, and how you were supported?
Personally, my career path was far from easy. I only managed to get my PhD at 38 years of age. I needed to first prove myself. Despite all my efforts and dozens of applications, I was never considered a good candidate. I needed to work for free for two years to land a proper job in my field of choice. During that time I took on many odd jobs to support myself. I worked for a top 10 university for free and they never saw my worth or gave me an opportunity. With that experience I landed a proper job at a leading pharmaceutical company. After one year with them, they funded my PhD studies and now here I am with a career in science.
Considering your own career route, what message do you have for people who would like to follow in your footsteps?
Never ever give up - it is possible. Look for the right mentors and be humble. You do not need to reinvent the wheel, but only to find someone who can lend you theirs. Learn to grow from the experiences of others and be ready to fail a couple of times - we all do. Be open to learn and never be afraid of following your dreams.
>> At SCI, we’re proud to support #BlackinChem. Reach out to us with your stories.
What do you think are the specific barriers that might be preventing young black people from pursuing chemistry/science?
I think one of the biggest barriers that prevent people from pursuing careers in science is the lack of role models. If we only show advertisements for chemistry degrees with White people, it’s not encouraging for Black students to pursue a career there. The same goes for when we visit universities; role models are needed. No one wants to be the only Black person in the department. Universities need to embrace diversity at all levels. I understand that tradition sometimes prevents this, but we need to change and ignore tradition for a bit.
What steps do you think can be taken by academia and businesses to increase the number of Black people studying and pursuing chemistry/science as a career?
Showcase Black chemists and inventors to motivate the younger generations and show society that Black people are not only artists and musicians. Target extracurricular activities in schools where children are from disadvantaged backgrounds. Train your staff to be open. Create cultural events that not only target Black people but also for other people to learn and see that in the end we are all equal. We all need to learn to embrace our differences and grow together.
>> As we celebrate #BlackinChem, we mark the achievements of some inspirational chemists. Read more about the amazing career of Percy Lavon Julian.
This week SCI is joining with business and academia to mark #BlackInChem, an initiative to advance and promote a new generation of Black chemists.
Over the coming weeks, we shall be profiling past and present Black chemists, many of whom are unsung heroes, and whose work established the foundations on which some of our modern science is built. We start with the outstanding contribution made by Percy Lavon Julian (1899-1975).
Born on 11 April 1899 in Montgomery, Alabama, US, Percy L Julian was the son of a clerk at the United State Post Office and a teacher. He did well at school, and even though there were no public high schools for African Americans in Montgomery, he was accepted at DePauw University, Indiana, in 1916.
Due to segregation Julian had to live off campus, even struggling initially to find somewhere that would serve him food. As well as completing his studies, he worked to pay his college expenses. Excelling in his studies, he graduated with a BA in 1920.
Julian wanted to study chemistry, but with little encouragement to continue his education, based on the fact there were few job opportunities, he found a position as a chemistry instructor at Fisk University, Nashville, Tennessee.
In 1922 Julian won an Austin Fellowship to Harvard University and received his MA in 1923. With no job offers forthcoming, he served on the staff of predominantly Black colleges, first at West Virginia State College and in 1928 as head of the department of chemistry at Howard University.
In 1929 Julian received a Rockefeller Foundation grant and the chance to earn his doctorate in chemistry. He studied natural products chemistry with Ernst Späth, an Austrian chemist, at the University of Vienna and received his PhD in 1931. He returned to Howard University, but it is said that internal politics forced him to leave.
Physostigmine was synthesised by Julian
Julian returned to DePauw University as a research fellow during 1933. Collaborating with fellow chemist and friend Josef Pikl, he completed research, in 1935, that resulted in the synthesis of physostigmine. His work was published in the Journal of the American Chemical Society.
Physostigmine, an alkaloid, was only available from its natural source, the Calabar bean, the seed of a leguminous plant native to tropical Africa. Julian’s research and synthesis process made the chemical readily available for the treatment of glaucoma. It is said that this development was the most significant chemical research publication to come from DePauw.
Calabar bean
Once the grant funding had expired, and despite efforts of those who championed his work, the Board of Trustees at DePauw would not allow Julian to be promoted to teaching staff. He left to pursue a distinguished career in industry. It is said that he was denied one particular position as a town law forbid ‘housing of a Negro overnight.’ Other companies are also said to have rejected him because of his race.
However, in 1936 he was offered a position as director of research for soya products at Glidden in Chicago. Over the next 18 years, the results of his soybean protein research produced numerous patents and successful products for Glidden. These included a paper coating and a fire-retardant foam used widely in World War II to extinguish gasoline fires. Julian’s biomedical research made it possible to produce large quantities of synthetic progesterone and hydrocortisone at low cost.
Percy Lavon Julian | Editorial credit: spatuletail / Shutterstock.com
By 1953 Julian Laboratories had been established, an enterprise that he went on to sell for more than $2 million in 1961. He then established the Julian Research Institute, a non-profit research organisation. In 1967 he was appointed to the DePauw University Board of Trustees, and in 1973 he was elected to the National Academy of Sciences, the second African American to receive the honour.
He was also widely recognised as a steadfast advocate of human rights. Julian continued his private research studies and served as a consultant to major pharmaceutical companies until his death on 19 April 1975. Percy Lavon Julian is commemorated at DePauw University with the Percy L Julian Science and Mathematics Center named in his honour. During 1993 the United States Postal Service commemorated Julian on a stamp in recognition of his extraordinary contribution to science and society.
From genome mining and green synthesis, to tackling tuberculosis and computational methods to help cure malaria, the chemists of tomorrow have been busy showcasing their talents as part of the Society of Chemical Industry Young Chemists Panel’s National Undergraduate Online Poster Competition 2021.
A snapshot of these students’ talents is bottled below in their own words. So, which one of these 15 entries do you think contains the most potential?
1: Genome mining for myxobacterial natural products discovery
Emmanuelle Acs et al., University of Glasgow
Natural products have always had a privileged place in drug development programmes, but their discovery is long and tedious. Genome mining arises as a solution allowing the finding of compounds never seen before. Using an array of bioinformatic softwares, the myxobacterial genome was explored for new Ribosomally and Post-Translationally modified Peptides (RIPPs). Myxobacteria are soil-dwelling bacteria known for the number of secondary metabolites they produce, and they have proven to hide many more within their genome. Indeed, our analyses have led to the potential discovery of nine new myxobacterial natural products. The nature and class of these products is to be confirmed by biosynthesis in the laboratory.
2: De novo design of a lanthanide-binding peptide inspired by the methanol dehydrogenase enzyme active site
Olivia Baldwin et al., University of Birmingham
Lanthanides were thought to be biologically irrelevant until the discovery of bacteria containing the lanthanide-dependent methanol dehydrogenase (Ln-MDH) enzyme. There has been interest in exploiting the attractive properties of the lanthanides by the de novo design of artificial proteins, aiming to explore protein structures and functions not observed in biology. Here, a lanthanide-binding peptide, CS1-0, has been designed de novo and shown to bind to europium and pyrroloquinoline-quinone (PQQ), a key component of the Ln-MDH active site. This partial recreation of a biologically relevant lanthanide binding site is a step towards the ultimate goal of de novo design, to create functional artificial metalloproteins with simplified structures.
3: Template-directed synthesis of porphyrin nanorings: a computational and experimental study
Janko Hergenhahn et al., University of Oxford
Template-directed synthesis provides a route to achieve porphyrin nanorings by favouring ring-closure reaction over oligomerisation. A structurally new template with 12 binding sites has been proposed for the synthesis of novel porphyrin rings; however, initial unsuccessful reactions have raised questions about the binding efficiency of this template to the linear substrate. We have employed classical and quantum modelling together with experimental techniques to explore template-substrate binding in solution and shed light on this process. Titration experiments and modelling have enabled us to study the occupancy of different binding sites and quantify the influence of strain on binding, further guiding novel designs.
4: Transborylation enabled boron-catalysed hydrocyanation of enones
Kieran Benn et al., University of Edinburgh
Hydrocyanation offers an orthogonal route to synthetically ubiquitous amines. Current hydrocyanation methodologies are dominated by the use of acutely toxic hydrogen cyanide gas and transition metal catalysts. Here the application of main-group catalysis and transborylation is reported for the formal hydrocyanation of functionalised alkenes. The catalytic protocol was optimised and applied to a broad range of substrates (20 examples), including examples where chemoselectivity was demonstrated in the presence of reducible functionalities and Lewis basic groups. Mechanistic studies support a proposed catalytic cycle in which B–N/B–H transborylation was a key to catalyst turnover.
Students at the University of Glasgow have used computational analysis to help tackle malaria.
5: Investigation of the structure-property relationship between hydrophobicity and the antimicrobial activity of AMP-mimic copolymers
Xiyue Leng et al., University of Birmingham
Antimicrobial peptides are increasingly employed as new-generation antibiotics, with their amphiphilic nature (contain both hydrophobic and cationic components) mimicked by polymers to enable a more cost-effective approach. However, there is a lack of a quantitative pre-experiment indicator to provide a prospect on their potency. The overall hydrophobicity represented by LogP/SA was proposed to rapidly identify candidates in future designing to reduce synthetic efforts. We show a comparison study between two computational tools used to calculate LogP/SA: ChemBio3D and Materials Studio, in terms of the predictive power and sensitivity, followed by the synthesis of copolymers with a different cationic side chain based on the calculation results.
6: Modelling potential SARS-CoV-2 VIRTAC warheads
Mirjam-Kim Rääbis et al., University of Glasgow
Traditional small molecule therapeutics in medicinal chemistry often require high doses to inhibit the target protein, leading to issues with safety and drug resistance. Proteolysis targeting chimeras (PROTACs) are a new class of molecule that combat these issues, as they can use the body’s own protein degradation systems to degrade targets even at low drug doses. Virus-targeting chimeras (VIRTACs) can use a similar mechanism to target viral proteins. This project uses molecular docking studies to explore potential VIRTAC warheads that target the papain-like protease of SARS-CoV-2, in an attempt to find a potential treatment to COVID-19 that would, among other benefits, offer a lower risk of antiviral resistance.
7: Design, synthesis and biological evaluation of anti-tubercular small molecules
Miriam Turner et al., Newcastle University
Tuberculosis remains one of the top 10 causes of death worldwide, therefore there exists an unmet clinical need for new and improved therapeutics that tackle increasing bacterial resistance and affordability issues. Previous studies indicate N-substituted amino acid hydrazides exhibit good activity against several strains of Mtb. Ongoing structure-activity relationship studies utilising isoniazid, a variety of amino acids, and the active imidazo[1,2-a]pyridine-3-carboxy moiety of clinical candidate Q203 have also demonstrated excellent activities. Herein we report the results of our continued evaluation of this architecture, using a scaffold hopping approach to explore the potential of this pharmacophore as a new anti-tubercular drug.
8: Towards a cure for malaria: computational analysis and design of PfCLK3 inhibitors
Skye Brettell et al., University of Glasgow
Malaria continues to pose a significant challenge to humanity. Resistance to several frontline antimalarials represents a considerable threat, marking the need for new drugs with novel mechanisms of action. Kinase inhibitors represent a potential new class of antimalarials. TCMDC-135051 is a hit compound with activity against malarial kinase PfCLK3 as well as potency in liver, blood and sexual stage parasites. During this project, sequential analysis of the PfCLK3 catalytic domain identified key structural differences between the target and its human orthologs. Molecular docking studies of TCMDC-135051 analogues using GOLD then yielded potential lead compounds with predicted high affinity for the target kinase.
9: Mind the gap! Through-space electronic stabilisation of dynamic azaacene-extended helicene diimides
Matteo Albino et al., University of York
The strain-induced contortion of non-planar, chiroptically-active helicenes caused by fjord steric repulsive interactions is well known. Fjord-mediated planarisation, on the other hand, is far less common and has typically only been achieved via inherently strong covalent bond formation. Herein, I present the properties and density functional theory (DFT) analysis of electroactive aza[5]helicenes exhibiting unexpected through-space π-electronic stabilisation in the reduced states as a result of non-covalent fjord bonding effects. Computational modelling of optical spectra and aromatic-induced current densities reveal that lone pair-repulsive nitrogens in the fjord promote favourable ring currents and reversible helicene planarisation.
10: Mitochondria targeted metal-chelators as potential therapeutic agents against Parkinson’s Disease
Sam Andrew Young et al., Northumbria University
The synthesis of metal chelating molecules, specifically hydroxypyridones (HOPOs), have been identified as potential therapeutic agents for treating Parkinson’s Disease (PD) as bidentate ligands at the two oxygen donor atoms. These ligands are selective for ferric iron in the body, which is expected to stop the reduction of this iron accumulated in the brains of PD sufferers, hindering the Haber-Weiss mechanism from taking place in the mitochondria of the cell and preventing the associated degeneration of the cells. The lipophilicity of these HOPOs is vital to the process, allowing the molecule to transverse the blood-brain barrier, the addition of a triphenylphosphonium group on the HOPO is thought to increase therapeutic effect.
At Heriot Watt University, students have investigated the skin irritation potential of nanoclays using an IATA
11: Green synthesis of n-acetylcolchinol
Adelaide Lunga et al., Loughborough University
The aim of this project is to develop a short synthesis of N-acetylcolchinol using a greener and step-economical pathway. First, aldol condensation of 3-hydroxyacetophenone and 3,4,5-trimethoxybenzaldehye using ethanolic NaOH produced the respective chalcone. The product was reduced electrochemically in DMSO:MeOH (4:1) employing carbon electrodes and NEt4Cl to the saturated benzylic alcohol, which was converted to an acetamide via Ritter reaction using H2SO4 in MeCN. In the final step, the conditions were optimised to enable electrochemical oxidative coupling of the aromatic groups to give the desired N-acetylcolchinol. This novel four-steps reaction sequence avoids use of transition metal catalysts or toxic reagents.
12: Towards the synthesis of small-molecule probes to measure endosulfatase activity
Yi Xiao et al., University of Oxford
Human endosulfatases (SULFs) are enzymes on the cell surface and in the extracellular matrix that hydrolyse 6-O-sulfate on glucosamine units within heparan sulfate proteoglycans. SULFs are involved in growth and development, muscle regeneration and tumour growth via various signaling pathways, with untapped therapeutic and diagnostic potentials. However, profiling SULFs remains a challenge. Antibodies detect their presence, but do not indicate their activity state. The current activity assay is a global sulfatase assay and is not selective in a biological sample. We propose a novel small-molecule probe to profile SULF activity by exploiting the formation of 1,6-anhydrosugar, which can be potentially used in isolated proteins and in vitro.
13: Developing machine learning models to predict the Hansen solubility parameters
Alexander Pine et al., University of Greenwich
Solubility parameters are important for pharmaceutical formulations, paint formulations and new material development. There is a need to improve the accuracy of solubility calculations, and to be able to make rapid predictions of the solubility of new molecular structures. In this project, a range of Python plugins, and open-source codes have been used to develop a Lasso linear regression machine learning model to predict the Hansen solubility parameters (HSP) - δd, δp and δh, which represents dispersion forces, dipole-permanent dipole forces and hydrogen bonding respectively with the intention of making faster and more accurate prediction in solubility.
14: The cycloaddition between cyclononyne and mesyl azide
Alexander David Robertson et al., The University of Glasgow
This research considers computational modelling of a SPAAC reaction involving cyclononyne. DFT calculations were performed on the strain promoted reaction between cyclononyne and mesyl azide. Three low energy conformers of cyclononyne with Cs, C2 and C1 symmetry were found with similar energy. The transition structures for the corresponding cycloaddition with mesyl azide were found and the C2 conformer was the lowest in energy. Product structures were found leading to the identification of the thermodynamic product of the reaction. Distortion/interaction analysis showed that the cycloalkyne was already significantly pre-constrained to its reacting geometry.
15: The assessment of skin irritation potential of nanoclays using an IATA
Holly King et al., Heriot Watt university
Clays are natural nanomaterials consisting of mineral silicate layers. They have several functional uses in everyday life. An example of nanoclays that carry out a wide range of roles is smectites which include montmorillonite (MMT), bentonite and hectorite. These nanoclays can be used in cosmetics, altering their appearance and in pharmaceuticals as drug carriers and wound dressings. Integrated approach to testing and assessment (IATA) aim to collect all relevant data into one easy to understand format that can be used to group materials. Using an IATA dedicated to skin irritation/corrosion it was found that MMT was safe for use. However, hectorite was found to be toxic at high doses indicating that it is a possible irritant to the skin.
Many thanks to the sponsors of this year’s competition: GSK, AstraZeneca, TeledyneIsco. The event runs until 9 July, so let us know what you think of the entries on Twitter at #SCIPosterComp.
If you’d like to see these students’ full posters, go to: https://istry.co.uk/postercompetition/5/?date_example=2021-06-28
Every tin can dropped into our recycling bins is a small act of faith. We hope each one is recycled, yet the figures take some of that fervour from our faith. According to UK government statistics from 2015-2018, only about 45% of our household waste is recycled. Similarly, the UN has noted that only 20% of the 50 million tonnes of electronics waste produced globally each year is formally recycled. So, it’s fair to say we could do better.
Thankfully, thousands of people around the globe are working on these problems and two recent developments give us grounds for optimism. The first involves upcycling metal waste into multi-purpose aerogels, and the second involves fully recyclable printed electronics that include a wood-derived ink.
Upcycling metal waste
Researchers at the National University of Singapore (NUS) claim to have turned one person’s trash into treasure with a low-energy way to convert aluminium and magnesium waste into high value aerogels for the construction industry.
To do this, they ground the metal waste into a powder and mixed it with chemical cross-linkers. They heated this mixture in an oven before freeze-drying it and turning it into an aerogel. The team says this simple process makes their aerogels 50% cheaper than commercially available silica aerogels.
Aerogels have many useful properties. They are absorbent, extremely light (hence the frozen smoke nickname), and have impressive thermal and sound insulation capabilities. This makes them useful as thermal insulation materials in buildings, in piping systems, or for cleaning up oil spills. However, the NUS team has loftier goals than that.
There is a great need for less energy intensive ways to recycle metals
“Our aluminium aerogel is 30 times lighter and insulates heat 21 times better than conventional concrete,” research team leader Associate Professor Duong Hai-Minh whose research has been published in the Journal of Material Cycles and Waste Management. “When optical fibres are added during the mixing stage, we can create translucent aluminium aerogels which, as building materials, can improve natural lighting, reduce energy consumption for lighting and illuminate dark or windowless areas. Translucent concrete can also be used to construct sidewalks and speed bumps that light up at night to improve safety for pedestrians and road traffic.”
The aerogels could even be used for cell cultivation. Professor Duong explains: “Microcarriers are micro-size beads for cells to anchor and grow. Our first trials were performed on stem cells, using a cell line commonly used for testing of drugs as well as cosmetics, and the results are very encouraging.”
Whatever about these speculative applications, this upcycling method will hopefully help us find new homes for all types of metal waste including metal chips and discarded electronics.
Recyclable printed electronics
A team at Duke University has also made interesting progress in reducing electronic waste. The researchers claim to have developed fully recyclable printed electronics that could be used and reused in a wide range of sensors.
The researchers’ transistor is made from three carbon-based inks that can be printed onto paper, and their use of a wood-derived insulating dielectric ink called nanocellulose helps make them recyclable. Carbon nanotubes and graphene inks are also used for the semiconductors and conductors, respectively.
A 3D rendering of the first fully recyclable, printed transistor. CREDIT: Duke University
“Nanocellulose is biodegradable and has been used in applications like packaging for years,” said Aaron Franklin, the Addy Professor of Electrical and Computer Engineering at Duke, whose research has been published in Nature Electronics. “And while people have long known about its potential applications as an insulator in electronics, nobody has figured out how to use it in a printable ink before. That’s one of the keys to making these fully recyclable devices functional.”
The team has developed a way to suspend these nanocellulose crystals (extracted from wood fibres) with a sprinkling of table salt to create an ink that performs well in its printed transistors. At the end of their working life, these devices can be submerged in baths with gently vibrating sound waves to recover the carbon nanotubes and graphene components. These materials can be reused and the nanocellulose can be recycled just like ordinary paper.
The team conceded that these devices won’t ruffle the trillion dollar silicon-based computer component market, but they do think these devices could be useful in simple environmental sensors to monitor building energy use or in biosensing patches to track medical conditions.
Read about the Duke University research here: https://www.nature.com/articles/s41928-021-00574-0
Take a look at the NUS study here: https://link.springer.com/article/10.1007/s10163-020-01169-1